Novel easily separable core-shell Fe3O4/PVP/ZIF-8 nanostructure adsorbent: optimization of phosphorus removal from Fosfomycin pharmaceutical wastewater
- PMID: 35496345
- PMCID: PMC9044422
- DOI: 10.1039/d2ra00936f
Novel easily separable core-shell Fe3O4/PVP/ZIF-8 nanostructure adsorbent: optimization of phosphorus removal from Fosfomycin pharmaceutical wastewater
Abstract
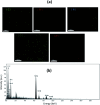
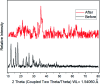
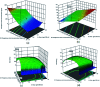
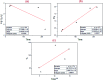
A new easily separable core-shell Fe3O4/PVP/ZIF-8 nanostructure adsorbent was synthesized and then examined for removal of Fosfomycin antibiotic from synthetic pharmaceutical wastewater. The removal process of Fosfomycin was expressed through testing the total phosphorus (TP). A response surface model (RSM) for Fosfomycin adsorption (as mg-P L-1) was used by carrying out the experiments using a central composite design. The adsorption model showed that Fosfomycin adsorption is directly proportional to core-shell Fe3O4/PVP/ZIF-8 nanostructure adsorbent dosage and time, and indirectly to initial Fosfomycin concentration. The removal increased by decreasing the pH to 2. The Fosfomycin removal was done at room temperature under an orbital agitation speed of 250 rpm. The adsorption capacity of core-shell Fe3O4/PVP/ZIF-8 nanostructure adsorbent reached around 1200 mg-P g-1, which is significantly higher than other MOF adsorbents reported in the literature. The maximum Langmuir adsorption capacity of the adsorbent for Fosfomycin was 126.58 mg g-1 and Fosfomycin adsorption behavior followed the Freundlich isotherm (R 2 = 0.9505) in the present study. The kinetics was best fitted by the pseudo-second-order model (R 2 = 0.9764). The RSM model was used for the adsorption process in different target modes.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures











References
-
- Alazaiza M. Y. D. Albahnasawi A. Ali G. A. M. Bashir M. J. K. Nassani D. E. Al Maskari T. Abu Amr S. S. Abujazar M. S. S. Application of Natural Coagulants for Pharmaceutical Removal from Water and Wastewater: A Review. Water. 2022;14:1–16. doi: 10.3390/w14020140. doi: 10.3390/w14020140. - DOI
-
- Buffle M. O. Schumacher J. Salhi E. Jekel M. von Gunten U. Measurement of the initial phase of ozone decomposition in water and wastewater by means of a continuous quench-flow system: Application to disinfection and pharmaceutical oxidation. Water Res. 2006;40:1884–1894. doi: 10.1016/j.watres.2006.02.026. doi: 10.1016/j.watres.2006.02.026. - DOI - PubMed
-
- Pérez-Estrada L. A. Malato S. Gernjak W. Agüera A. Thurman E. M. Ferrer I. Fernández-Alba A. R. Photo-fenton degradation of diclofenac: Identification of main intermediates and degradation pathway. Environ. Sci. Technol. 2005;39:8300–8306. doi: 10.1021/es050794n. doi: 10.1021/es050794n. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

