Mitoapocynin Attenuates Organic Dust Exposure-Induced Neuroinflammation and Sensory-Motor Deficits in a Mouse Model
- PMID: 35496912
- PMCID: PMC9043522
- DOI: 10.3389/fncel.2022.817046
Mitoapocynin Attenuates Organic Dust Exposure-Induced Neuroinflammation and Sensory-Motor Deficits in a Mouse Model
Abstract
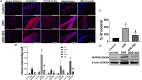
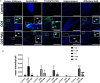
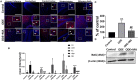
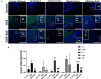
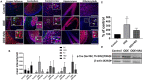
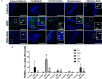
Increased incidences of neuro-inflammatory diseases in the mid-western United States of America (USA) have been linked to exposure to agriculture contaminants. Organic dust (OD) is a major contaminant in the animal production industry and is central to the respiratory symptoms in the exposed individuals. However, the exposure effects on the brain remain largely unknown. OD exposure is known to induce a pro-inflammatory phenotype in microglial cells. Further, blocking cytoplasmic NOX-2 using mitoapocynin (MA) partially curtail the OD exposure effects. Therefore, using a mouse model, we tested a hypothesis that inhaled OD induces neuroinflammation and sensory-motor deficits. Mice were administered with either saline, fluorescent lipopolysaccharides (LPSs), or OD extract intranasally daily for 5 days a week for 5 weeks. The saline or OD extract-exposed mice received either a vehicle or MA (3 mg/kg) orally for 3 days/week for 5 weeks. We quantified inflammatory changes in the upper respiratory tract and brain, assessed sensory-motor changes using rotarod, open-field, and olfactory test, and quantified neurochemicals in the brain. Inhaled fluorescent LPS (FL-LPS) was detected in the nasal turbinates and olfactory bulbs. OD extract exposure induced atrophy of the olfactory epithelium with reduction in the number of nerve bundles in the nasopharyngeal meatus, loss of cilia in the upper respiratory epithelium with an increase in the number of goblet cells, and increase in the thickness of the nasal epithelium. Interestingly, OD exposure increased the expression of HMGB1, 3- nitrotyrosine (NT), IBA1, glial fibrillary acidic protein (GFAP), hyperphosphorylated Tau (p-Tau), and terminal deoxynucleotidyl transferase deoxyuridine triphosphate (dUTP) nick end labeling (TUNEL)-positive cells in the brain. Further, OD exposure decreased time to fall (rotarod), total distance traveled (open-field test), and olfactory ability (novel scent test). Oral MA partially rescued olfactory epithelial changes and gross congestion of the brain tissue. MA treatment also decreased the expression of HMGB1, 3-NT, IBA1, GFAP, and p-Tau, and significantly reversed exposure induced sensory-motor deficits. Neurochemical analysis provided an early indication of depressive behavior. Collectively, our results demonstrate that inhalation exposure to OD can cause sustained neuroinflammation and behavior deficits through lung-brain axis and that MA treatment can dampen the OD-induced inflammatory response at the level of lung and brain.
Keywords: inflammation; microglia; mitoapocynin; neurodegeneration; organic dust.
Copyright © 2022 Massey, Shrestha, Bhat, Padhi, Wang, Karriker, Smith, Kanthasamy and Charavaryamath.
Conflict of interest statement
AK is a shareholder of PK Biosciences Corporation Ames, IA, which is interested in identifying novel biomarkers and potential therapeutic targets for Parkinson's disease. The remaining authors declare that they have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures


Similar articles
-
HMGB1-RAGE Signaling Plays a Role in Organic Dust-Induced Microglial Activation and Neuroinflammation.Toxicol Sci. 2019 Jun 1;169(2):579-592. doi: 10.1093/toxsci/kfz071. Toxicol Sci. 2019. PMID: 30859215 Free PMC article.
-
Organic dust-induced mitochondrial dysfunction could be targeted via cGAS-STING or cytoplasmic NOX-2 inhibition using microglial cells and brain slice culture models.Cell Tissue Res. 2021 May;384(2):465-486. doi: 10.1007/s00441-021-03422-x. Epub 2021 Mar 9. Cell Tissue Res. 2021. PMID: 33687557 Free PMC article.
-
NTP Toxicology and Carcinogenesis Studies of Isobutyraldehyde (CAS No. 78-84-2) in F344/N Rats and B6C3F1 Mice (Inhalation Studies).Natl Toxicol Program Tech Rep Ser. 1999 Feb;472:1-242. Natl Toxicol Program Tech Rep Ser. 1999. PMID: 12579201
-
Toxicology and carcinogenesis studies of propargyl alcohol (CAS No. 107-19-7) in F344/N rats and B6C3F1 mice (inhalation studies).Natl Toxicol Program Tech Rep Ser. 2008 Sep;(552):1-172. Natl Toxicol Program Tech Rep Ser. 2008. PMID: 18974778
-
NTP Toxicology and Carcinogenesis Studies of Nickel Sulfate Hexahydrate (CAS No. 10101-97-0) in F344 Rats and B6C3F1 Mice (Inhalation Studies).Natl Toxicol Program Tech Rep Ser. 1996 Jul;454:1-380. Natl Toxicol Program Tech Rep Ser. 1996. PMID: 12587012
Cited by
-
Neuroinflammaging: A Tight Line Between Normal Aging and Age-Related Neurodegenerative Disorders.Aging Dis. 2024 Aug 1;15(4):1726-1747. doi: 10.14336/AD.2023.1001. Aging Dis. 2024. PMID: 38300639 Free PMC article. Review.
-
Decoding mechanisms and protein markers in lung-brain axis.Respir Res. 2025 May 19;26(1):190. doi: 10.1186/s12931-025-03272-z. Respir Res. 2025. PMID: 40390067 Free PMC article. Review.
-
Life-threatening risk factors contribute to the development of diseases with the highest mortality through the induction of regulated necrotic cell death.Cell Death Dis. 2025 Apr 11;16(1):273. doi: 10.1038/s41419-025-07563-7. Cell Death Dis. 2025. PMID: 40216765 Free PMC article. Review.
References
-
- Cacciottolo M., Wang X., Driscoll I., Woodward N., Saffari A., Reyes J., et al. . (2017). Particulate air pollutants, APOE alleles and their contributions to cognitive impairment in older women and to amyloidogenesis in experimental models. Transl. Psychiatry. 7, e1022. 10.1038/tp.2016.280 - DOI - PMC - PubMed
-
- Calderón-Garcidueñas L., Kavanaugh M., Block M., D'Angiulli A., Delgado-Chávez R., Torres-Jardón R., et al. . (2012). Neuroinflammation, hyperphosphorylated tau, diffuse amyloid plaques, and down-regulation of the cellular prion protein in air pollution exposed children and young adults. J. Alzheimer's Dis. 28, 93–107. 10.3233/JAD-2011-110722 - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

