Need for split: integrative taxonomy reveals unnoticed diversity in the subaquatic species of Pseudohygrohypnum (Pylaisiaceae, Bryophyta)
- PMID: 35497188
- PMCID: PMC9053303
- DOI: 10.7717/peerj.13260
Need for split: integrative taxonomy reveals unnoticed diversity in the subaquatic species of Pseudohygrohypnum (Pylaisiaceae, Bryophyta)
Abstract
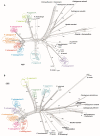
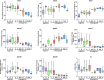
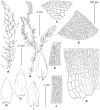
We present an integrative molecular and morphological study of subaquatic representatives of the genus Pseudohygrohypnum (Pylaisiaceae, Bryophyta), supplemented by distribution modelling of the revealed phylogenetic lineages. Phylogenetic analyses of nuclear and plastid datasets combined with the assemble species by automatic partitioning (ASAP) algorithm revealed eight distinct species within the traditionally circumscribed P. eugyrium and P. subeugyrium. These species are therefore yet another example of seemingly widely distributed taxa that harbour molecularly well-differentiated lineages with narrower distribution ranges. Studied accessions that were previously assigned to P. eugyrium form three clearly allopatric lineages, associated with temperate regions of Europe, eastern North America and eastern Asia. Remarkably, accessions falling under the current morphological concept of P. subeugyrium were shown to be even more diverse, containing five phylogenetic lineages. Three of these lineages occur under harsh Asian continental climates from cool-temperate to Arctic regions, while the remaining two, referred to P. subeugyrium s.str. and P. purpurascens, have more oceanic North Atlantic and East Asian distributions. Niche identity and similarity tests suggested no similarity in the distributions of the phylogenetically related lineages but revealed the identity of two East Asian species and the similarity of two pairs of unrelated species. A morphological survey confirmed the distinctness of all eight phylogenetic lineages, requiring the description of five new species. Pseudohygrohypnum appalachianum and P. orientale are described for North American and East Asian plants of P. eugyrium s.l., while P. sibiricum, P. subarcticum and P. neglectum are described for the three continental, predominantly Asian lineages of P. subeugyrium s.l. Our results highlight the importance of nontropical Asia as a center of bryophyte diversity. Phylogenic dating suggests that the diversification of subaquatic Pseudohygrohypnum lineages appeared in late Miocene, while mesophilous species of the genus split before Miocene cooling, in climatic conditions close to those where the ancestor of Pseudohygrohypnum appeared. We speculate that radiation of the P. subeugyrium complex in temperate Asia might have been driven by progressive cooling, aridification, and increases in seasonality, temperature and humidity gradients. Our results parallel those of several integrative taxonomic studies of North Asian mosses, which have resulted in a number of newly revealed species. These include various endemics from continental areas of Asia suggesting that the so-called Rapoport's rule of low diversity and wide distribution range in subpolar regions might not be applicable to bryophytes. Rather, the strong climatic oscillations in these regions may have served as a driving force of speciation and niche divergence.
Keywords: Allopatry; Biogeography; Cryptic diversity; Disjunctive distribution; Hypnales; ITS; North Asia; Species distribution model; trnS-trnF.
©2022 Fedosov et al.
Conflict of interest statement
The authors declare there are no competing interests.
Figures
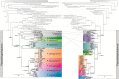













Similar articles
-
Disentangling phylogenetic relations and biogeographic history within the Cucujus haematodes species group (Coleoptera: Cucujidae).Mol Phylogenet Evol. 2022 Aug;173:107527. doi: 10.1016/j.ympev.2022.107527. Epub 2022 May 14. Mol Phylogenet Evol. 2022. PMID: 35577286
-
Do mosses really exhibit so large distribution ranges? Insights from the integrative taxonomic study of the Lewinskya affinis complex (Orthotrichaceae, Bryopsida).Mol Phylogenet Evol. 2019 Nov;140:106598. doi: 10.1016/j.ympev.2019.106598. Epub 2019 Aug 17. Mol Phylogenet Evol. 2019. PMID: 31430552
-
Phylogeography reveals an ancient cryptic radiation in East-Asian tree frogs (Hyla japonica group) and complex relationships between continental and island lineages.BMC Evol Biol. 2016 Nov 23;16(1):253. doi: 10.1186/s12862-016-0814-x. BMC Evol Biol. 2016. PMID: 27884104 Free PMC article.
-
Molecular phylogeny reveals distinct evolutionary lineages of the banded krait, Bungarus fasciatus (Squamata, Elapidae) in Asia.Sci Rep. 2023 Feb 4;13(1):2061. doi: 10.1038/s41598-023-28241-8. Sci Rep. 2023. PMID: 36739450 Free PMC article. Review.
-
Global advances in phylogeny, taxonomy and biogeography of Lauraceae.Plant Divers. 2025 Apr 7;47(3):341-364. doi: 10.1016/j.pld.2025.04.001. eCollection 2025 May. Plant Divers. 2025. PMID: 40496996 Free PMC article. Review.
Cited by
-
On the Cryptic Speciation in the Mosses with East Asia-East North America Disjunction: A Case Study of Two Poorly Understood Mosses from the Southern Extremity of the Russian Far East.Plants (Basel). 2024 Dec 20;13(24):3558. doi: 10.3390/plants13243558. Plants (Basel). 2024. PMID: 39771256 Free PMC article.
-
Integrative Taxonomy Reveals Hidden Diversity in the Aloina catillum Complex (Pottiaceae, Bryophyta).Plants (Basel). 2024 Feb 2;13(3):445. doi: 10.3390/plants13030445. Plants (Basel). 2024. PMID: 38337977 Free PMC article.
-
New Taxonomic Arrangement of Dicranella s.l. and Aongstroemia s.l. (Dicranidae, Bryophyta).Plants (Basel). 2023 Mar 17;12(6):1360. doi: 10.3390/plants12061360. Plants (Basel). 2023. PMID: 36987046 Free PMC article.
References
-
- Afonina OM. On moss flora of the Sokhondinsky State Nature Biosphere Reserve (Zabaikalsky Territory) Arctoa. 2009;18:141–150. doi: 10.15298/arctoa.18.07. - DOI
-
- Allen BH. Maine Mosses: Drummondiaceae—Polytrichaceae. Memoirs of the New York Botanical Garden. 2014;111:1–607.
-
- Anderson LE, Zander RH. Mosses of the southern Blue Ridge province and their phytogeographic relationships. http://www.jstor.org/stable/24333419 Journal of the Elisha Mitchell Scientific Society. 1973;89(1/2):15–60.
-
- Bakalin VA, Vilnet AA. Two new species of the liverwort genus Hygrobiella Spruce (Marchantiophyta) described from the North Pacific based on integrative taxonomy. Plant Systematics and Evolution. 2014;300:2277–2291. doi: 10.1007/s00606-014-1050-8. - DOI
Further Reading
-
- Zomer RJ, Trabucco A, Bossio DA, Verchot LV. Climate change mitigation: a spatial analysis of global land suitability for clean development mechanism afforestation and reforestation. Agriculture, Ecosystems & Environment. 2008;126(1–2):67–80. doi: 10.1016/j.agee.2008.01.014. - DOI
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Research Materials

