Iron-dependent epigenetic modulation promotes pathogenic T cell differentiation in lupus
- PMID: 35499082
- PMCID: PMC9057600
- DOI: 10.1172/JCI152345
Iron-dependent epigenetic modulation promotes pathogenic T cell differentiation in lupus
Abstract
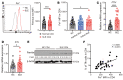
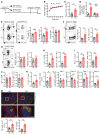
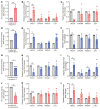
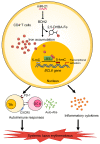
The trace element iron affects immune responses and vaccination, but knowledge of its role in autoimmune diseases is limited. Expansion of pathogenic T cells, especially T follicular helper (Tfh) cells, has great significance to systemic lupus erythematosus (SLE) pathogenesis. Here, we show an important role of iron in regulation of pathogenic T cell differentiation in SLE. We found that iron overload promoted Tfh cell expansion, proinflammatory cytokine secretion, and autoantibody production in lupus-prone mice. Mice treated with a high-iron diet exhibited an increased proportion of Tfh cell and antigen-specific GC response. Iron supplementation contributed to Tfh cell differentiation. In contrast, iron chelation inhibited Tfh cell differentiation. We demonstrated that the miR-21/BDH2 axis drove iron accumulation during Tfh cell differentiation and further promoted Fe2+-dependent TET enzyme activity and BCL6 gene demethylation. Thus, maintaining iron homeostasis might be critical for eliminating pathogenic Th cells and might help improve the management of patients with SLE.
Keywords: Autoimmunity; Epigenetics; Lupus; T cells.
Figures






Comment in
-
Labile iron accumulation augments T follicular helper cell differentiation.J Clin Invest. 2022 May 2;132(9):e159472. doi: 10.1172/JCI159472. J Clin Invest. 2022. PMID: 35499081 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

