In Vivo Multi-Day Calcium Imaging of CA1 Hippocampus in Freely Moving Rats Reveals a High Preponderance of Place Cells with Consistent Place Fields
- PMID: 35501152
- PMCID: PMC9172072
- DOI: 10.1523/JNEUROSCI.1750-21.2022
In Vivo Multi-Day Calcium Imaging of CA1 Hippocampus in Freely Moving Rats Reveals a High Preponderance of Place Cells with Consistent Place Fields
Abstract
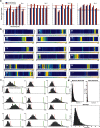
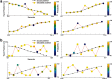
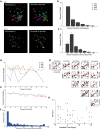
Calcium imaging using GCaMP indicators and miniature microscopes has been used to image cellular populations during long timescales and in different task phases, as well as to determine neuronal circuit topology and organization. Because the hippocampus (HPC) is essential for tasks of memory, spatial navigation, and learning, calcium imaging of large populations of HPC neurons can provide new insight on cell changes over time during these tasks. All reported HPC in vivo calcium imaging experiments have been done in mouse. However, rats have many behavioral and physiological experimental advantages over mice. In this paper, we present the first (to our knowledge) in vivo calcium imaging from CA1 HPC in freely moving male rats. Using the UCLA Miniscope, we demonstrate that, in rat, hundreds of cells can be visualized and held across weeks. We show that calcium events in these cells are highly correlated with periods of movement, with few calcium events occurring during periods without movement. We additionally show that an extremely large percent of cells recorded during a navigational task are place cells (77.3 ± 5.0%, surpassing the percent seen during mouse calcium imaging), and that these cells enable accurate decoding of animal position and can be held over days with consistent place fields in a consistent spatial map. A detailed protocol is included, and implications of these advancements on in vivo imaging and place field literature are discussed.SIGNIFICANCE STATEMENT In vivo calcium imaging in freely moving animals allows the visualization of cellular activity across days. In this paper, we present the first in vivo Ca2+ recording from CA1 hippocampus (HPC) in freely moving rats. We demonstrate that hundreds of cells can be visualized and held across weeks, and that calcium activity corresponds to periods of movement. We show that a high percentage (77.3 ± 5.0%) of imaged cells are place cells, and that these place cells enable accurate decoding and can be held stably over days with little change in field location. Because the HPC is essential for many tasks involving memory, navigation, and learning, imaging of large populations of HPC neurons can shed new insight on cellular activity changes and organization.
Keywords: Ca1; calcium imaging; hippocampus; miniscopes; place cells; rats.
Copyright © 2022 the authors.
Figures



References
-
- Ahanonu B (2018) CIAtah: a software package for analyzing one- and two-photon calcium imaging datasets, v1.0.0 Edition. Zenodo.
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
