Role of cathepsin K in the expression of mechanical hypersensitivity following intra-plantar inflammation
- PMID: 35501334
- PMCID: PMC9061763
- DOI: 10.1038/s41598-022-11043-9
Role of cathepsin K in the expression of mechanical hypersensitivity following intra-plantar inflammation
Abstract
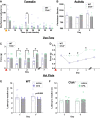
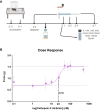
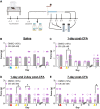
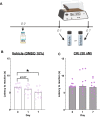
Persistent/chronic inflammatory pain involves multiple pathophysiological mechanisms and is far more complex than acute/momentary pain. Current therapeutics for chronic inflammatory pain are often not effective because the etiology responsible for the pain is not addressed by traditional pharmacological treatments. Cathepsin K is a cysteine protease that has mostly been studied in the context of bone and joint disorders. Previous work by others has shown that inhibition of cathepsin K activity reduces osteoarthritis-associated nociception in joints. However, the role of cathepsin K in cutaneous inflammation is understudied. We assessed the effectiveness of genetic deletion or pharmacological inhibition of cathepsin K in male mice on the expression of nocifensive behaviors after formalin injection or mechanical and thermal hypersensitivity after injection of complete Freund's adjuvant (CFA) into the mouse hind paw. Our data demonstrate that cathepsin K knockout mice (Ctsk-/-) have a reduction in nocifensive behaviors in the formalin test. In addition, Ctsk-/- do not develop mechanical hypersensitivity after CFA injection for up to 7 days. Moreover, we found that inhibition of cathepsin K reduced mechanical hypersensitivity after CFA injection and mRNA levels, protein levels, and cathepsin K activity levels were elevated after CFA injection. Based upon our data, cathepsin K is indicated to play a role in the expression of chemically-induced cutaneous hypersensitivity, as Ctsk-/- mice do not develop mechanical hypersensitivity and show a reduction in nocifensive behaviors. Further research is needed to determine whether attenuating cathepsin K activity may generate a clinically relevant therapeutic.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures

References
-
- Cenac N. In: Proteases and Their Receptors in Inflammation Progress in Inflammation Research. Chignard N, Vergnolle M, editors. Springer; 2011. pp. 253–274.
-
- Feizerfan A, Sheh G. Transition from acute to chronic pain. Contin. Educ. Anaesth. Crit. Care Pain. 2015;15:98–102. doi: 10.1093/bjaceaccp/mku044. - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

