Treatment with IFB-088 Improves Neuropathy in CMT1A and CMT1B Mice
- PMID: 35501630
- PMCID: PMC9167212
- DOI: 10.1007/s12035-022-02838-y
Treatment with IFB-088 Improves Neuropathy in CMT1A and CMT1B Mice
Abstract
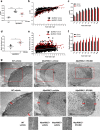
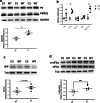
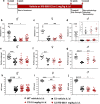
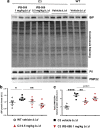
Charcot-Marie-Tooth disease type 1A (CMT1A), caused by duplication of the peripheral myelin protein 22 (PMP22) gene, and CMT1B, caused by mutations in myelin protein zero (MPZ) gene, are the two most common forms of demyelinating CMT (CMT1), and no treatments are available for either. Prior studies of the MpzSer63del mouse model of CMT1B have demonstrated that protein misfolding, endoplasmic reticulum (ER) retention and activation of the unfolded protein response (UPR) contributed to the neuropathy. Heterozygous patients with an arginine to cysteine mutation in MPZ (MPZR98C) develop a severe infantile form of CMT1B which is modelled by MpzR98C/ + mice that also show ER stress and an activated UPR. C3-PMP22 mice are considered to effectively model CMT1A. Altered proteostasis, ER stress and activation of the UPR have been demonstrated in mice carrying Pmp22 mutations. To determine whether enabling the ER stress/UPR and readjusting protein homeostasis would effectively treat these models of CMT1B and CMT1A, we administered Sephin1/IFB-088/icerguestat, a UPR modulator which showed efficacy in the MpzS63del model of CMT1B, to heterozygous MpzR98C and C3-PMP22 mice. Mice were analysed by behavioural, neurophysiological, morphological and biochemical measures. Both MpzR98C/ + and C3-PMP22 mice improved in motor function and neurophysiology. Myelination, as demonstrated by g-ratios and myelin thickness, improved in CMT1B and CMT1A mice and markers of UPR activation returned towards wild-type values. Taken together, our results demonstrate the capability of IFB-088 to treat a second mouse model of CMT1B and a mouse model of CMT1A, the most common form of CMT. Given the recent benefits of IFB-088 treatment in amyotrophic lateral sclerosis and multiple sclerosis animal models, these data demonstrate its potential in managing UPR and ER stress for multiple mutations in CMT1 as well as in other neurodegenerative diseases. (Left panel) the accumulation of overexpressed PMP22 or misfolded mutant P0 in the Schwann cell endoplasmic reticulum (ER) leads to overwhelming of the degradative capacity, activation of ER-stress mechanisms, and myelination impairment. (Right panel) by prolonging eIF2α phosphorylation, IFB-088 reduces the amount of newly synthesized proteins entering the ER, allowing the protein quality control systems to better cope with the unfolded/misfolded protein and allowing myelination to progress.
Keywords: Charcot-Marie-Tooth; IFB-088/Sephin1/icerguestat; Neuropathy; Proteostasis; UPR.
© 2022. The Author(s).
Conflict of interest statement
P.G., P.M. and C.T. are full-time employees and stockholders of InFlectis BioScience. M.D. acts as a Scientific Advisory Board member and Consultant for InFlectis BioScience.
Figures



References
-
- Fridman V, Bundy B, Reilly MM, Pareyson D, Bacon C, Burns J, Day J, Feely S, et al. CMT subtypes and disease burden in patients enrolled in the Inherited Neuropathies Consortium natural history study: a cross-sectional analysis. J Neurol Neurosurg Psychiatry. 2015;86:873–878. doi: 10.1136/jnnp-2014-308826. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

