Smartphone-based mobile applications for adverse drug reactions reporting: global status and country experience
- PMID: 35501745
- PMCID: PMC9063059
- DOI: 10.1186/s12911-022-01832-7
Smartphone-based mobile applications for adverse drug reactions reporting: global status and country experience
Abstract
Background: Smartphone technology can support paperless reporting of adverse drug reactions (ADRs). The aims of this study were to systematically assess smartphone ADR-reporting applications, understand their qualitative and quantitative impact on ADR reporting, and garner key lessons from owners and developers.
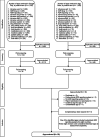
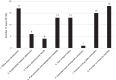
Methods: This study had three components: (1) An assessment of ADR-reporting apps, (2) an online survey on the impact of app implementation on ADR reporting and the experiences of app developers and owners, and (3) a search of VigiBase, the World Health Organization global database of individual case safety reports (ICSRs), to observe trends in the number of ADR reports targeting countries where the apps were implemented.
Results: Twenty-two apps were included. Eight out of the 22 apps were for countries in the WHO African region. Features observed included E2B data elements (E stands for efficacy) and functions supporting reporting and user engagement. Seventeen app developers and owners answered to the survey and reported overall positive experiences with app features, and post-launch increases in the total number of ICSRs. User type and user environment were cited as factors influencing app use: Respondents said younger people and/or those with an inclination to use technology were more likely to use apps compared to older or more technology-averse people, while respondents in countries with limited internet connectivity reported persistent difficulties in app use.
Conclusions: Smartphone apps for reporting ADRs offer added value compared to conventional reporting tools. Reporting tools should be selected based on interface features and factors that may influence app usage.
Keywords: Adverse drug reactions (ADRs); Drug safety; Pharmacovigilance; Regulatory system; Reporting of ADRs; Smartphone apps; VigiBase.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
-
- International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use: Post-Approval Safety Data Management: Definitions and Standards for Expedited Reporting E2D. https://database.ich.org/sites/default/files/E2D_Guideline.pdf.
-
- World Health Organization: Safety monitoring of medical products: reporting system for the general public; 2012.
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

