Direct conversion of cellulose to L-lactic acid by a novel thermophilic Caldicellulosiruptor strain
- PMID: 35501875
- PMCID: PMC9063331
- DOI: 10.1186/s13068-022-02137-7
Direct conversion of cellulose to L-lactic acid by a novel thermophilic Caldicellulosiruptor strain
Abstract
Background: Consolidated bioprocessing (CBP) of lignocellulosic biomass to L-lactic acid using thermophilic cellulolytic/hemicellulolytic bacteria provides a promising solution for efficient lignocellulose conversion without the need for additional cellulolytic/hemicellulolytic enzymes. Most studies on the mesophilic and thermophilic CBP of lignocellulose to lactic acid concentrate on cultivation of non-cellulolytic mesophilic and thermophilic bacteria at temperatures of 30-55 °C with external addition of cellulases/hemicellulases for saccharification of substrates.
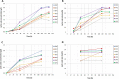
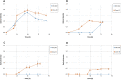
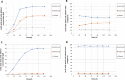
Results: L-Lactic acid was generated by fermenting microcrystalline cellulose or lignocellulosic substrates with a novel thermophilic anaerobic bacterium Caldicellulosiruptor sp. DIB 104C without adding externally produced cellulolytic/hemicellulolytic enzymes. Selection of this novel bacterium strain for lactic acid production is described as well as the adaptive evolution towards increasing the L-lactic acid concentration from 6 to 70 g/l on microcrystalline cellulose. The evolved strains grown on microcrystalline cellulose show a maximum lactic acid production rate of 1.0 g/l*h and a lactic acid ratio in the total organic fermentation products of 96 wt%. The enantiomeric purity of the L-lactic acid generated is 99.4%. In addition, the lactic acid production by these strains on several other types of cellulose and lignocellulosic feedstocks is also reported.
Conclusions: The evolved strains originating from Caldicellulosiruptor sp. DIB 104C were capable of producing unexpectedly large amounts of L-lactic acid from microcrystalline cellulose in fermenters. These strains produce L-lactic acid also from lignocellulosic feedstocks and thus represent an ideal starting point for development of a highly integrated commercial L-lactic acid production process from such feedstocks.
Keywords: Anaerobic; Caldicellulosiruptor; Consolidated bioprocessing; High temperature; L-Lactic acid; Lignocellulose; Thermophilic bacteria.
© 2022. The Author(s).
Conflict of interest statement
All authors are current employees of BluCon Biotech GmbH. BluCon actively develops processes for lignocellulose conversion to chemicals and fuels.
Figures

Similar articles
-
Single-step ethanol production from lignocellulose using novel extremely thermophilic bacteria.Biotechnol Biofuels. 2013 Feb 28;6(1):31. doi: 10.1186/1754-6834-6-31. Biotechnol Biofuels. 2013. PMID: 23448304 Free PMC article.
-
Single-step bioconversion of lignocellulose to hydrogen using novel moderately thermophilic bacteria.Biotechnol Biofuels. 2014 Jun 3;7:82. doi: 10.1186/1754-6834-7-82. eCollection 2014. Biotechnol Biofuels. 2014. PMID: 24920960 Free PMC article.
-
Cellulosic ethanol production via consolidated bioprocessing by a novel thermophilic anaerobic bacterium isolated from a Himalayan hot spring.Biotechnol Biofuels. 2017 Mar 21;10:73. doi: 10.1186/s13068-017-0756-6. eCollection 2017. Biotechnol Biofuels. 2017. PMID: 28344648 Free PMC article.
-
Metabolic engineering of Caldicellulosiruptor bescii for hydrogen production.Appl Microbiol Biotechnol. 2024 Dec;108(1):65. doi: 10.1007/s00253-023-12974-7. Epub 2024 Jan 9. Appl Microbiol Biotechnol. 2024. PMID: 38194138 Free PMC article. Review.
-
Endowing non-cellulolytic microorganisms with cellulolytic activity aiming for consolidated bioprocessing.Biotechnol Adv. 2013 Nov;31(6):754-63. doi: 10.1016/j.biotechadv.2013.02.007. Epub 2013 Mar 6. Biotechnol Adv. 2013. PMID: 23473971 Review.
Cited by
-
Metabolic engineering of Zymomonas mobilis for co-production of D-lactic acid and ethanol using waste feedstocks of molasses and corncob residue hydrolysate.Front Bioeng Biotechnol. 2023 Feb 21;11:1135484. doi: 10.3389/fbioe.2023.1135484. eCollection 2023. Front Bioeng Biotechnol. 2023. PMID: 36896016 Free PMC article.
-
The potential of native and engineered Clostridia for biomass biorefining.Front Bioeng Biotechnol. 2024 Aug 16;12:1423935. doi: 10.3389/fbioe.2024.1423935. eCollection 2024. Front Bioeng Biotechnol. 2024. PMID: 39219620 Free PMC article. Review.
-
A novel SfaNI-like restriction-modification system in Caldicellulosiruptor extents the genetic engineering toolbox for this genus.PLoS One. 2022 Dec 29;17(12):e0279562. doi: 10.1371/journal.pone.0279562. eCollection 2022. PLoS One. 2022. PMID: 36580476 Free PMC article.
-
The Undeniable Potential of Thermophiles in Industrial Processes.Int J Mol Sci. 2024 Jul 13;25(14):7685. doi: 10.3390/ijms25147685. Int J Mol Sci. 2024. PMID: 39062928 Free PMC article. Review.
-
Adaptive Evolution for the Efficient Production of High-Quality d-Lactic Acid Using Engineered Klebsiella pneumoniae.Microorganisms. 2024 Jun 8;12(6):1167. doi: 10.3390/microorganisms12061167. Microorganisms. 2024. PMID: 38930549 Free PMC article.
References
-
- Tan J, Abdel-Rahman MA, Sonomoto K. Biorefinery-based lactic acid fermentation: microbial production of pure monomer product. Adv Polym Sci. 2018;279:27–66. doi: 10.1007/12_2016_11. - DOI
LinkOut - more resources
Full Text Sources
