Gene Therapy for Fibrodysplasia Ossificans Progressiva: Feasibility and Obstacles
- PMID: 35502479
- PMCID: PMC9419966
- DOI: 10.1089/hum.2022.023
Gene Therapy for Fibrodysplasia Ossificans Progressiva: Feasibility and Obstacles
Abstract
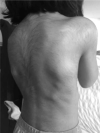
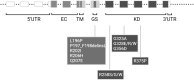
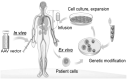
Fibrodysplasia ossificans progressiva (FOP) is a rare and devastating genetic disease, in which soft connective tissue is converted into heterotopic bone through an endochondral ossification process. Patients succumb early as they gradually become trapped in a second skeleton of heterotopic bone. Although the underlying genetic defect is long known, the inherent complexity of the disease has hindered the discovery of effective preventions and treatments. New developments in the gene therapy field have motivated its consideration as an attractive therapeutic option for FOP. However, the immune system's role in FOP activation and the as-yet unknown primary causative cell, are crucial issues which must be taken into account in the therapy design. While gene therapy offers a potential therapeutic solution, more knowledge about FOP is needed to enable its optimal and safe application.
Keywords: ALK2 mutation; RNA; fibrodysplasia ossificans progressiva; gene therapy; heterotopic ossification.
Conflict of interest statement
E.M.W.E.: PI clinical trials of Regeneron Pharmaceuticals, indirect EU-IMI by Astra Zeneca; Clementia Pharmaceuticals (an Ipsen Company). G.G. and J.-H.S.: funding sources are NIAMS of the NIH under R21AR077557, the AAVAA Therapeutics, IFOPA. F.S.K.: research investigator: Clementia/Ipsen, Regeneron. R.J.P.: R.J.P. is an investigator on FOP clinical trials sponsored by Clementia Pharmaceuticals (an Ipsen Company) and Regeneron Pharmaceuticals. R.J.P is a consultant for Ipsen, Regeneron, Incyte, Biocryst, and Keros, but receives no personal compensation for these activities. N.H.: Past Research Investigator: Clementia/Ipsen. Z.G. consultant and speaker for Ipsen BiopharmaceuticalsInc. Regeneron. C.L.D.C.: Clementia/Ipsen PI, Novartis: speaker, Biogen: speaker, Novo Nordisk: speaker. M.D.R. Disclosures last 2 years: Maja Di Rocco received fees for lectures or participation on advisory board by Sanofi, Takeda, Ultragenix, Orchard. Participating in FOP trials. P.D.: PI for Ipsen Move trial. T.-J.C. Funding source: research grant of the Genome Technology to Business Translation Program from the National Research Foundation (NRF) funded by the Ministry of Science, ICT & Future Planning of the Republic of Korea Government. E.H.: ECH is an investigator on FOP clinical trials sponsored by Clementia Pharmaceuticals (an Ipsen Company). No other conflict of interest related to this article.
Figures
References
-
- Kaplan FS, Tabas JA, Gannon FH, et al. The histopathology of fibrodysplasia ossificans progressiva. An endochondral process. J Bone Joint Surg Am 1993;75:220–230. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

