Recent Developments in Plasmonic Alloy Nanoparticles: Synthesis, Modelling, Properties and Applications
- PMID: 35502819
- PMCID: PMC9804694
- DOI: 10.1002/cphc.202200136
Recent Developments in Plasmonic Alloy Nanoparticles: Synthesis, Modelling, Properties and Applications
Abstract
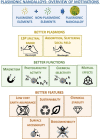
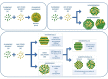
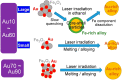
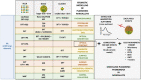
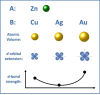
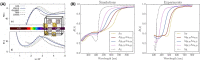
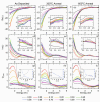
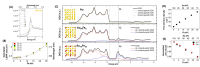
Despite the traditional plasmonic materials are counted on one hand, there are a lot of possible combinations leading to alloys with other elements of the periodic table, in particular those renowned for magnetic or catalytic properties. It is not a surprise, therefore, that nanoalloys are considered for their ability to open new perspectives in the panorama of plasmonics, representing a leading research sector nowadays. This is demonstrated by a long list of studies describing multiple applications of nanoalloys in photonics, photocatalysis, sensing and magneto-optics, where plasmons are combined with other physical and chemical phenomena. In some remarkable cases, the amplification of the conventional properties and even new effects emerged. However, this field is still in its infancy and several challenges must be overcome, starting with the synthesis (control of composition, crystalline order, size, processability, achievement of metastable phases and disordered compounds) as well as the modelling of the structure and properties (accuracy of results, reliability of structural predictions, description of disordered phases, evolution over time) of nanoalloys. To foster the research on plasmonic nanoalloys, here we provide an overview of the most recent results and developments in the field, organized according to synthetic strategies, modelling approaches, dominant properties and reported applications. Considering the several plasmonic nanoalloys under development as well as the large number of those still awaiting synthesis, modelling, properties assessment and technological exploitation, we expect a great impact on the forthcoming solutions for sustainability, ultrasensitive and accurate detection, information processing and many other fields.
Keywords: alloy nanoparticles; bimetallic nanoparticles; density functional calculations; photocatalysis; plasmon resonance.
© 2022 The Authors. ChemPhysChem published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
















References
-
- C. Kittel, Introduction to Solid State Physics, Wiley, 2005.
-
- Amendola V., Pilot R., Frasconi M., Maragò O. M., Iatì M. A., J. Phys. Condens. Matter 2017, 29, 203002. - PubMed
-
- Mayer K. M., Hafner J. H., Chem. Rev. 2011, 111, 3828. - PubMed
-
- Murphy C. J., Gole A. M., Stone J. W., Sisco P. N., Alkilany A. M., Goldsmith E. C., Baxter S. C., Acc. Chem. Res. 2008, 41, 1721. - PubMed
-
- Martirez J. M. P., Bao J. L., Carter E. A., Annu. Rev. Phys. Chem. 2021, 72, 061020. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

