177Lu-DOTA-0-Tyr3-octreotate infusion modeling for real-time detection and characterization of extravasation during PRRT
- PMID: 35503186
- PMCID: PMC9065226
- DOI: 10.1186/s40658-022-00466-y
177Lu-DOTA-0-Tyr3-octreotate infusion modeling for real-time detection and characterization of extravasation during PRRT
Abstract
Purpose: Given the recent and rapid development of peptide receptor radionuclide therapy (PRRT), increasing emphasis should be placed on the early identification and quantification of therapeutic radiopharmaceutical (thRPM) extravasation during intravenous administration. Herein, we provide an analytical model of 177Lu-DOTA0-Tyr3-octreotate (Lutathera®) infusion for real-time detection and characterization of thRPM extravasation.
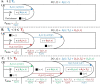
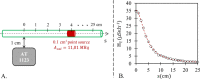
Methods: For 33 Lutathera®-based PRRT procedures using the gravity infusion method, equivalent dose rates (EDRs) were monitored at the patient's arm. Models of flow dynamics for nonextravasated and extravasated infusions were elaborated and compared to experimental data through an equivalent dose rate calibration. Nonextravasated infusion was modeled by assuming constant volume dilution of 177Lu activity concentration in the vial and Poiseuille-like laminar flow through the tubing and patient vein. Extravasated infusions were modeled according to their onset times by considering elliptically shaped extravasation region with different aspect ratios.
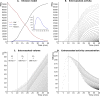
Results: Over the 33 procedures, the peak of the median EDR was reached 14 min after the start of the infusion with a value of 450 µSv h-1. On the basis of experimental measurements, 1 mSv h-1 was considered the empirical threshold for Lutathera® extravasation requiring cessation of the infusion and start again with a new route of injection. According to our model, the concentration of extravascular activity was directly related to the time of extravasation onset and its duration, a finding inherent in the gravity infusion method. This result should be considered when planning therapeutic strategy in the case of RPM extravasation because the local absorbed dose for β-emitters is closely linked to activity concentration. For selected EDR values, charts of extravasated activity, volume, and activity concentration were computed for extravasation characterization.
Conclusion: We proposed an analytical model of Lutathera® infusion and extravasation (gravity method) based on EDR monitoring. This approach could be useful for the early detection of thRPM extravasation and for the real-time assessment of activity concentration and volume accumulation in the extravascular medium.
Keywords: 177Lu; Equivalent dose rate; Extravasation; Gravity infusion method; Lutathera; Neuroendocrine tumors; Peptide receptor radionuclide therapy; Radioprotection.
© 2022. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures





References
-
- Tylski P, Vuillod A, Goutain-Majorel C, Jalade P. 58 Dose estimation for an extravasation in a patient treated with 177 Lu-DOTATATE. Phys Med. 2018;56:32–33. doi: 10.1016/j.ejmp.2018.09.071. - DOI
LinkOut - more resources
Full Text Sources
Miscellaneous

