The making of suberin
- PMID: 35510799
- PMCID: PMC9994434
- DOI: 10.1111/nph.18202
The making of suberin
Abstract
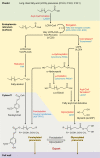
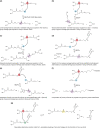
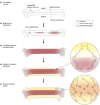
Outer protective barriers of animals use a variety of bio-polymers, based on either proteins (e.g. collagens), or modified sugars (e.g. chitin). Plants, however, have come up with a particular solution, based on the polymerisation of lipid-like precursors, giving rise to cutin and suberin. Suberin is a structural lipophilic polyester of fatty acids, glycerol and some aromatics found in cell walls of phellem, endodermis, exodermis, wound tissues, abscission zones, bundle sheath and other tissues. It deposits as a hydrophobic layer between the (ligno)cellulosic primary cell wall and plasma membrane. Suberin is highly protective against biotic and abiotic stresses, shows great developmental plasticity and its chemically recalcitrant nature might assist the sequestration of atmospheric carbon by plants. The aim of this review is to integrate the rapidly accelerating genetic and cell biological discoveries of recent years with the important chemical and structural contributions obtained from very diverse organisms and tissue layers. We critically discuss the order and localisation of the enzymatic machinery synthesising the presumed substrates for export and apoplastic polymerisation. We attempt to explain observed suberin linkages by diverse enzyme activities and discuss the spatiotemporal relationship of suberin with lignin and ferulates, necessary to produce a functional suberised cell wall.
Keywords: apoplastic barrier; cell wall; fatty acyl metabolism; ferulic acid; lignin; lipid intracellular transport; suberin; suberin lamellae.
© 2022 The Authors. New Phytologist © 2022 New Phytologist Foundation.
Figures

References
-
- Alejandro S, Lee Y, Tohge T, Sudre D, Osorio S, Park J, Bovet L, Lee Y, Geldner N, Fernie A et al. 2012. AtABCG29 is a monolignol transporter involved in lignin biosynthesis. Current Biology 22: 1207–1212. - PubMed
-
- An Q, Hückelhoven R, Kogel K‐H, Bel AJEV. 2006. Multivesicular bodies participate in a cell wall‐associated defence response in barley leaves attacked by the pathogenic powdery mildew fungus. Cellular Microbiology 8: 1009–1019. - PubMed
-
- Andersen TG, Molina D, Kilian J, Franke RB, Ragni L, Geldner N. 2021. Tissue‐autonomous phenylpropanoid production is essential for establishment of root barriers. Current Biology 31: 965–977. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

