Autophagy induced by taurolidine protects against polymicrobial sepsis by promoting both host resistance and disease tolerance
- PMID: 35512102
- PMCID: PMC9171638
- DOI: 10.1073/pnas.2121244119
Autophagy induced by taurolidine protects against polymicrobial sepsis by promoting both host resistance and disease tolerance
Abstract
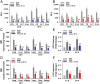
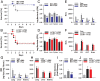
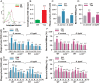
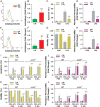
Sepsis, septic shock, and their sequelae are the leading causes of death in intensive care units, with limited therapeutic options. Disease resistance and tolerance are two evolutionarily conserved yet distinct defense strategies that protect the host against microbial infection. Here, we report that taurolidine administered at 6 h before septic challenge led to strong protection against polymicrobial sepsis by promoting both host resistance and disease tolerance characterized by accelerated bacterial clearance, ameliorated organ damage, and diminished vascular and gut permeability. Notably, taurolidine administered at 6 h after septic challenge also rescued mice from sepsis-associated lethality by enhancing disease tolerance to tissue and organ injury. Importantly, this in vivo protection afforded by taurolidine depends on an intact autophagy pathway, as taurolidine protected wild-type mice but was unable to rescue autophagy-deficient mice from microbial sepsis. In vitro, taurolidine induced light chain 3-associated phagocytosis in innate phagocytes and autophagy in vascular endothelium and gut epithelium, resulting in augmented bactericidal activity and enhanced cellular tolerance to endotoxin-induced damage in these cells. These results illustrate that taurolidine-induced autophagy augments both host resistance and disease tolerance to bacterial infection, thereby conferring protection against microbial sepsis.
Keywords: autophagy; disease tolerance; host resistance; microbial sepsis.
Conflict of interest statement
The authors declare no competing interest.
Figures



Similar articles
-
Taurolidine, an antilipopolysaccharide agent, has immunoregulatory properties that are mediated by the amino acid taurine.J Leukoc Biol. 1995 Sep;58(3):299-306. doi: 10.1002/jlb.58.3.299. J Leukoc Biol. 1995. PMID: 7665985
-
Deletion of Nlrp3 Augments Survival during Polymicrobial Sepsis by Decreasing Autophagy and Enhancing Phagocytosis.J Immunol. 2017 Feb 1;198(3):1253-1262. doi: 10.4049/jimmunol.1601745. Epub 2016 Dec 28. J Immunol. 2017. PMID: 28031338 Free PMC article.
-
Effect of the antiendotoxic agent, taurolidine, in the treatment of sepsis syndrome: a placebo-controlled, double-blind trial.Crit Care Med. 1995 Jun;23(6):1033-9. doi: 10.1097/00003246-199506000-00007. Crit Care Med. 1995. PMID: 7774213 Clinical Trial.
-
Redox-directed cancer therapeutics: Taurolidine and Piperlongumine as broadly effective antineoplastic agents (review).Int J Oncol. 2014 Oct;45(4):1329-36. doi: 10.3892/ijo.2014.2566. Epub 2014 Jul 28. Int J Oncol. 2014. PMID: 25175943 Free PMC article. Review.
-
The evolving role of taurolidine in cancer therapy.Ann Surg Oncol. 2010 Apr;17(4):1135-43. doi: 10.1245/s10434-009-0867-9. Epub 2009 Dec 29. Ann Surg Oncol. 2010. PMID: 20039217 Review.
Cited by
-
Autophagy and autophagic cell death in sepsis: friend or foe?J Intensive Care. 2024 Oct 25;12(1):41. doi: 10.1186/s40560-024-00754-y. J Intensive Care. 2024. PMID: 39449054 Free PMC article. Review.
-
Braun Lipoprotein Protects against Escherichia coli-Induced Inflammatory Responses and Lethality in Mice.Microbiol Spectr. 2023 Mar 14;11(2):e0354122. doi: 10.1128/spectrum.03541-22. Online ahead of print. Microbiol Spectr. 2023. PMID: 36916913 Free PMC article.
-
Investigating the differential microRNAs expression in young and aged Drosophila melanogaster following Flock House Virus infection.Virulence. 2025 Dec;16(1):2549497. doi: 10.1080/21505594.2025.2549497. Epub 2025 Aug 25. Virulence. 2025. PMID: 40852959 Free PMC article.
-
Sepsis-Induced Endothelial Dysfunction: Permeability and Regulated Cell Death.J Inflamm Res. 2024 Nov 28;17:9953-9973. doi: 10.2147/JIR.S479926. eCollection 2024. J Inflamm Res. 2024. PMID: 39628705 Free PMC article. Review.
-
Effect of taurine supplementation on preventing ventilator-associated pneumonia in pediatrics under mechanical ventilation, a randomized controlled double-blind clinical trial.Front Pediatr. 2025 Jan 10;12:1490247. doi: 10.3389/fped.2024.1490247. eCollection 2024. Front Pediatr. 2025. PMID: 39867697 Free PMC article.
References
-
- Angus D. C., van der Poll T., Severe sepsis and septic shock. N. Engl. J. Med. 369, 840–851 (2013). - PubMed
-
- Nguyen H. B., Smith D., Sepsis in the 21st century: Recent definitions and therapeutic advances. Am. J. Emerg. Med. 25, 564–571 (2007). - PubMed
-
- Hotchkiss R. S., Karl I. E., The pathophysiology and treatment of sepsis. N. Engl. J. Med. 348, 138–150 (2003). - PubMed
-
- Martin G. S., Mannino D. M., Eaton S., Moss M., The epidemiology of sepsis in the United States from 1979 through 2000. N. Engl. J. Med. 348, 1546–1554 (2003). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

