CXCL16/ERK1/2 pathway regulates human podocytes growth, migration, apoptosis and epithelial mesenchymal transition
- PMID: 35514316
- PMCID: PMC9133949
- DOI: 10.3892/mmr.2022.12728
CXCL16/ERK1/2 pathway regulates human podocytes growth, migration, apoptosis and epithelial mesenchymal transition
Abstract
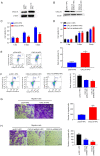
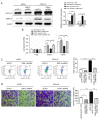
Primary nephrotic syndrome (PNS) is the commonest glomerular disease affecting children. Previous studies have confirmed that CXC motif chemokine ligand 16 (CXCL16) is involved in the pathogenesis of PNS. However, the exact mechanisms underlying the pathogenesis of PNS remain to be elucidated. Thus, the present study aimed to elucidate the role of CXCL16 in PNS. It was found that the expression of CXCL16 and extracellular signal‑regulated kinases 1 and 2 (ERK1/2) were significantly increased in clinical PNS renal tissues using reverse transcription‑quantitative PCR, western blot analysis and immunohistochemistry. Lentivirus overexpression or short hairpin RNA vector was used to induce the overexpression or knockdown of CXCL16 in podocytes, respectively. Overexpression of CXCL16 in podocytes could decrease the cell proliferation and increase the migration and apoptosis, whereas CXCL16 knockdown increased cell proliferation and decreased cell migration and apoptosis. Results of the present study further demonstrated that ERK2 protein expression was regulated by CXCL16. The knockdown of ERK2 expression reversed the effects of CXCL16 on the proliferation, apoptosis, migration and epithelial mesenchymal transition (EMT) of podocytes. Collectively, the findings of the present study highlighted that the CXCL16/ERK1/2 pathway regulates the growth, migration, apoptosis and EMT of human podocytes.
Keywords: CXC motif chemokine ligand 16; epithelial mesenchymal transition; extracellular signal‑regulated kinases 1 and 2; podocytes injury; primary nephrotic syndrome.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures



Similar articles
-
oxLDL promotes podocyte migration by regulating CXCL16, ADAM10 and ACTN4.Mol Med Rep. 2020 Sep;22(3):1976-1984. doi: 10.3892/mmr.2020.11292. Epub 2020 Jun 30. Mol Med Rep. 2020. PMID: 32705248 Free PMC article.
-
Circ_0000524/miR-500a-5p/CXCL16 axis promotes podocyte apoptosis in membranous nephropathy.Eur J Clin Invest. 2021 Mar;51(3):e13414. doi: 10.1111/eci.13414. Epub 2020 Oct 15. Eur J Clin Invest. 2021. PMID: 32974919
-
CXCL16 Promotes Gastric Cancer Tumorigenesis via ADAM10-Dependent CXCL16/CXCR6 Axis and Activates Akt and MAPK Signaling Pathways.Int J Biol Sci. 2021 Jul 5;17(11):2841-2852. doi: 10.7150/ijbs.57826. eCollection 2021. Int J Biol Sci. 2021. PMID: 34345211 Free PMC article.
-
P2X7 receptor inhibition attenuates podocyte injury by oxLDL through deregulating CXCL16.Cell Biol Int. 2022 Mar;46(3):454-461. doi: 10.1002/cbin.11742. Epub 2021 Dec 28. Cell Biol Int. 2022. PMID: 34882892
-
Parthenolide ameliorates tweak-induced podocytes injury.Mol Biol Rep. 2020 Jul;47(7):5165-5173. doi: 10.1007/s11033-020-05591-4. Epub 2020 Jun 22. Mol Biol Rep. 2020. PMID: 32572732
Cited by
-
Curcumin targets CXCL16-mediated podocyte injury and lipid accumulation in diabetic kidney disease treatment.Arch Pharm Res. 2024 Dec;47(12):924-939. doi: 10.1007/s12272-024-01521-1. Epub 2024 Nov 28. Arch Pharm Res. 2024. PMID: 39607647
-
The role of ERK1/2 signaling in diabetes: pathogenic and therapeutic implications.Front Pharmacol. 2025 May 9;16:1600251. doi: 10.3389/fphar.2025.1600251. eCollection 2025. Front Pharmacol. 2025. PMID: 40417223 Free PMC article. Review.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

