Azaheterocyclic diphenylmethanol chiral solvating agents for the NMR chiral discrimination of alpha-substituted carboxylic acids
- PMID: 35514411
- PMCID: PMC9056771
- DOI: 10.1039/d0ra06312f
Azaheterocyclic diphenylmethanol chiral solvating agents for the NMR chiral discrimination of alpha-substituted carboxylic acids
Abstract
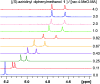
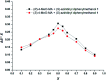
A series of small-membered heterocycle probes, so-called azaheterocycle-containing diphenylmethanol chiral solvating agents (CSAs), have been developed for NMR enantiodiscrimination. These chiral sensors were readily synthesized were inexpensive and efficiently used for the chiral analysis of alpha-substituted carboxylic acids. The sensing method was operationally simple and the processing was straightforward. Notably, we propose (S)-aziridinyl diphenylmethanol as a promising CSA, which has excellent chiral discriminating properties and offers multiple detectable possibilities pertaining to the 1H NMR signals of diagnostic split protons (including 25 examples, up to 0.194 ppm, 77.6 Hz). Its ability to detect the molecular recognition of fluorinated carboxylic acids were further investigated, with a good level of discrimination via the 19F NMR spectroscopic analysis. In addition, an accurate enantiomeric excess (ee) analysis of the p-methoxyl-mandelic acid with different optical compositions have been calculated based on the integration of well-separated proton signals.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures


Similar articles
-
DACH-Based Chiral Sensing Platforms as Tunable Benzamide-Chiral Solvating Agents for NMR Enantioselective Discrimination.Anal Chem. 2025 Jan 28;97(3):1900-1908. doi: 10.1021/acs.analchem.4c06024. Epub 2025 Jan 14. Anal Chem. 2025. PMID: 39810460
-
Efficient Enantiodifferentiation of Carboxylic Acids Using BINOL-Based Amino Alcohol as a Chiral NMR Solvating Agent.Front Chem. 2020 May 4;8:336. doi: 10.3389/fchem.2020.00336. eCollection 2020. Front Chem. 2020. PMID: 32432082 Free PMC article.
-
A chiral bisthiourea as a chiral solvating agent for carboxylic acids in the presence of DMAP.J Org Chem. 2013 Sep 20;78(18):9137-42. doi: 10.1021/jo4013546. Epub 2013 Sep 5. J Org Chem. 2013. PMID: 24050150
-
Determination of Enantiomeric Excess via 31P-NMR.Chirality. 2025 Apr;37(4):e70032. doi: 10.1002/chir.70032. Chirality. 2025. PMID: 40170346 Review.
-
Recent Advances in Multinuclear NMR Spectroscopy for Chiral Recognition of Organic Compounds.Molecules. 2017 Feb 7;22(2):247. doi: 10.3390/molecules22020247. Molecules. 2017. PMID: 28178223 Free PMC article. Review.
Cited by
-
A Squaramide-Based Organocatalyst as a Novel Versatile Chiral Solvating Agent for Carboxylic Acids.Molecules. 2024 May 19;29(10):2389. doi: 10.3390/molecules29102389. Molecules. 2024. PMID: 38792248 Free PMC article.
-
Unveiling the Power of Computational Tools in Chiral Liquid Chromatography.Molecules. 2025 Jul 31;30(15):3218. doi: 10.3390/molecules30153218. Molecules. 2025. PMID: 40807393 Free PMC article. Review.
-
Chiral Recognition of Chiral (Hetero)Cyclic Derivatives Probed by Tetraaza Macrocyclic Chiral Solvating Agents via 1H NMR Spectroscopy.Anal Chem. 2024 Apr 2;96(13):5188-5194. doi: 10.1021/acs.analchem.3c05395. Epub 2024 Mar 20. Anal Chem. 2024. PMID: 38506628 Free PMC article.
-
Chiral Recognition of Hydantoin Derivatives Enabled by Tetraaza Macrocyclic Chiral Solvating Agents Using 1H NMR Spectroscopy.J Org Chem. 2022 Jun 17;87(12):7934-7944. doi: 10.1021/acs.joc.2c00587. Epub 2022 Jun 8. J Org Chem. 2022. PMID: 35675642 Free PMC article.
-
Atropisomeric Racemization Kinetics of MRTX1719 Using Chiral Solvating Agent-Assisted 19F NMR Spectroscopy.ACS Omega. 2022 Aug 31;7(36):32062-32067. doi: 10.1021/acsomega.2c03316. eCollection 2022 Sep 13. ACS Omega. 2022. PMID: 36120049 Free PMC article.
References
-
-
For recent reviews on chiral analysis, see:
- You L. Zha D. Anslyn E. V. Chem. Rev. 2015;115:7840. doi: 10.1021/cr5005524. - DOI - PubMed
- Chen Z. Wang Q. Wu X. Li Z. Jiang Y. B. Chem. Soc. Rev. 2015;44:4249. doi: 10.1039/C4CS00531G. - DOI - PubMed
- Shen J. Okamoto Y. Chem. Rev. 2016;116:1094. doi: 10.1021/acs.chemrev.5b00317. - DOI - PubMed
-
-
-
For select reviews on analytical methods, see:
- Izake E. L. J. Pharm. Sci. 2007;96:1659. doi: 10.1002/jps.20820. - DOI - PubMed
- Okamoto Y. Ikai T. Chem. Soc. Rev. 2008;37:2593. doi: 10.1039/B808881K. - DOI - PubMed
- Pu L. Chem. Rev. 2004;104:1687. doi: 10.1021/cr030052h. - DOI - PubMed
- Jo H. H. Lin C. Y. Anslyn E. V. Acc. Chem. Res. 2014;47:2212. doi: 10.1021/ar500147x. - DOI - PubMed
-
-
- Seco J. M. Quinoa E. Riguera R. Chem. Rev. 2004;104:17. doi: 10.1021/cr000665j. - DOI - PubMed
- Wenzel T. J. Chisholm C. D. Prog. NMR Spectrosc. 2011;59:1. doi: 10.1016/j.pnmrs.2010.07.003. - DOI - PubMed
- Xu Z. Liu C. Zhao S. Chen S. Zhao Y. Chem. Rev. 2019;119:195. doi: 10.1021/acs.chemrev.8b00202. - DOI - PubMed
- Wenzel T. J., Discrimination of Chiral Compounds Using NMR Spectroscopy, John Wiley & Sons, Inc., 2nd edn, 2018, pp. 441–481
- Balzano F., Uccello-Barretta G., and Aiello F., Chiral Analysis by NMR Spectroscopy: Chiral Solvating Agents, Elsevier B.V., 2018, pp. 367–427
-
-
For select recent examples, see:
- Zhao Y. Swager T. M. J. Am. Chem. Soc. 2015;137:3221. doi: 10.1021/jacs.5b00556. - DOI - PMC - PubMed
- Seo M. S. Kim H. J. Am. Chem. Soc. 2015;137:14190. doi: 10.1021/jacs.5b09555. - DOI - PubMed
- Li L. P. Ye B. H. Inorg. Chem. 2017;56:10717. doi: 10.1021/acs.inorgchem.7b01681. - DOI - PubMed
- Aizawa S. I. Okano M. Kidani T. Chirality. 2017;29:273. doi: 10.1002/chir.22681. - DOI - PubMed
- Jang S. Kim H. iScience. 2019;19:425. doi: 10.1016/j.isci.2019.07.051. - DOI - PMC - PubMed
- Sun Z. Chen Z. Wang Y. Zhang X. Xu J. Bian G. Song L. Org. Lett. 2020;22:589. doi: 10.1021/acs.orglett.9b04373. - DOI - PubMed
- Jang S. Kim H. Org. Lett. 2020;22:4185. doi: 10.1021/acs.orglett.0c01256. - DOI - PubMed
-
-
-
For select recent examples, see:
- Couffin A. Boullay O. T. d. Vedrenne M. Navarro C. Martin-Vaca B. Bourissou D. Chem. Commun. 2014;50:5997. doi: 10.1039/C4CC00466C. - DOI - PubMed
- Jain N. Mandal M. B. Bedekar A. V. Tetrahedron. 2014;70:4343. doi: 10.1016/j.tet.2014.05.001. - DOI
- Bian G. Fan H. Huang H. Yang S. Zong H. Song L. Yang G. Org. Lett. 2015;17:1369. doi: 10.1021/acs.orglett.5b00030. - DOI - PubMed
- Monteagudo E. March P. Alvarez-Larena A. Virgili A. ChemistrySelect. 2017;2:7362. doi: 10.1002/slct.201701429. - DOI
- Khanvilkar A. Bedekar A. Chem. Commun. 2018;54:11037. doi: 10.1039/C8CC06245E. - DOI - PubMed
- Cuřínová P. Hájek P. Janků K. Holakovský R. Chirality. 2019;31:410. doi: 10.1002/chir.23067. - DOI - PubMed
- Prasad D. Mogurampelly S. Chaudhari S. R. RSC Adv. 2020;10:2303. doi: 10.1039/C9RA07803G. - DOI - PMC - PubMed
- Balzano F. Uccello-Barretta G. RSC Adv. 2020;10:4869. doi: 10.1039/D0RA00200C. - DOI - PMC - PubMed
- Recchimurzo A. Micheletti C. Uccello-Barretta G. Balzano F. J. Org. Chem. 2020;85:5342. doi: 10.1021/acs.joc.0c00027. - DOI - PMC - PubMed
-
LinkOut - more resources
Full Text Sources

