A Nicotinamide Phosphoribosyltransferase Inhibitor, FK866, Suppresses the Growth of Anaplastic Meningiomas and Inhibits Immune Checkpoint Expression by Regulating STAT1
- PMID: 35515130
- PMCID: PMC9065474
- DOI: 10.3389/fonc.2022.836257
A Nicotinamide Phosphoribosyltransferase Inhibitor, FK866, Suppresses the Growth of Anaplastic Meningiomas and Inhibits Immune Checkpoint Expression by Regulating STAT1
Abstract
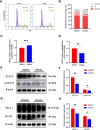
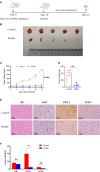
Anaplastic meningioma is classified as a World Health Organization (WHO) grade III tumor and shows a strong tendency to recur. Although the incidence of anaplastic meningioma is low, the high rate of recurrence and death still makes treatment a challenge. A proteomics analysis was performed to investigate the differentially expressed proteins between anaplastic meningiomas and fibrous meningiomas by micro-LC-MS/MS. The key metabolic enzyme nicotinamide phosphoribosyltransferase (NAMPT) showed upregulated expression in anaplastic meningiomas. However, targeting NAMPT to treat anaplastic meningiomas has not been reported. In vitro, NAMPT inhibitor -FK866 reduced the viability of anaplastic meningiomas by inducing cell cycle arrest at the G2/M phase. Intriguingly, the NAMPT inhibitor -FK866 decreased the protein expression of immune checkpoints PD-L1 and B7-H3 by down-regulating the STAT1 and p-STAT1 expression in vitro. Furthermore, FK866 suppressed the growth of anaplastic meningiomas in an in vivo xenograft model. The expression of Ki-67 and immune checkpoint proteins (PD-L1 and B7-H3) showed significant differences between the group treated with FK866 and the control group treated with DMSO. In conclusion, the expression of NAMPT, which plays a crucial role in energy metabolism, was upregulated in anaplastic meningiomas. The NAMPT inhibitor -FK866 significantly suppressed the growth of anaplastic meningiomas in vitro and in vivo. More strikingly, FK866 potently inhibited immune checkpoint protein (PD-L1 and B7-H3) expression by regulating STAT1 in vitro and in vivo. Our results demonstrated that NAMPT inhibitors could potentially be an effective treatment method for patients suffering from anaplastic meningiomas.
Keywords: NAMPT; NAMPT inhibitor; anaplastic meningioma; cell cycle; immune checkpoint; proteomics.
Copyright © 2022 Deng, Hu, Miao, Wang, Zhang, Wan, Wu, Lv, Feng, Ji, Park and Hao.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
The NAMPT Inhibitor FK866 in Combination with Cisplatin Reduces Cholangiocarcinoma Cells Growth.Cells. 2023 Feb 28;12(5):775. doi: 10.3390/cells12050775. Cells. 2023. PMID: 36899911 Free PMC article.
-
NAD+ Metabolic Enzyme Inhibitor as Radiosensitizer for Malignant Meningioma and its Modulation of P53 Expression.Mol Cancer Ther. 2024 Nov 4;23(11):1586-1596. doi: 10.1158/1535-7163.MCT-23-0632. Mol Cancer Ther. 2024. PMID: 39039948
-
Inhibiting PP2A Upregulates B7-H3 Expression and Potentially Increases the Sensitivity of Malignant Meningiomas to Immunotherapy by Proteomics.Pathol Oncol Res. 2022 Sep 20;28:1610572. doi: 10.3389/pore.2022.1610572. eCollection 2022. Pathol Oncol Res. 2022. PMID: 36203966 Free PMC article.
-
Clinical significance of checkpoint regulator "Programmed death ligand-1 (PD-L1)" expression in meningioma: review of the current status.J Neurooncol. 2021 Feb;151(3):443-449. doi: 10.1007/s11060-020-03584-8. Epub 2021 Feb 21. J Neurooncol. 2021. PMID: 33611710 Free PMC article. Review.
-
Medicinal chemistry of nicotinamide phosphoribosyltransferase (NAMPT) inhibitors.J Med Chem. 2013 Aug 22;56(16):6279-96. doi: 10.1021/jm4001049. Epub 2013 May 31. J Med Chem. 2013. PMID: 23679915 Review.
Cited by
-
Channeling Nicotinamide Phosphoribosyltransferase (NAMPT) to Address Life and Death.J Med Chem. 2024 Apr 25;67(8):5999-6026. doi: 10.1021/acs.jmedchem.3c02112. Epub 2024 Apr 5. J Med Chem. 2024. PMID: 38580317 Free PMC article. Review.
-
B7-H3 in Brain Malignancies: Immunology and Immunotherapy.Int J Biol Sci. 2023 Jul 24;19(12):3762-3780. doi: 10.7150/ijbs.85813. eCollection 2023. Int J Biol Sci. 2023. PMID: 37564196 Free PMC article. Review.
-
Synthesis and Evaluation of Reactive Oxygen Species Sensitive Prodrugs of a NAMPT Inhibitor FK866.Molecules. 2022 Dec 25;28(1):169. doi: 10.3390/molecules28010169. Molecules. 2022. PMID: 36615364 Free PMC article.
-
Meningioma animal models: a systematic review and meta-analysis.J Transl Med. 2023 Oct 28;21(1):764. doi: 10.1186/s12967-023-04620-7. J Transl Med. 2023. PMID: 37898750 Free PMC article.
-
The NAMPT Inhibitor FK866 in Combination with Cisplatin Reduces Cholangiocarcinoma Cells Growth.Cells. 2023 Feb 28;12(5):775. doi: 10.3390/cells12050775. Cells. 2023. PMID: 36899911 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

