Lipofection with estrone-based luminophores featuring aggregation-induced emission properties
- PMID: 35515436
- PMCID: PMC9054052
- DOI: 10.1039/d0ra03608k
Lipofection with estrone-based luminophores featuring aggregation-induced emission properties
Abstract
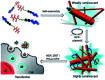
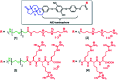
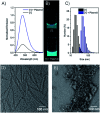
In this communication we present the use of a novel class of luminophores with aggregation-induced emission (AIE) properties based on the steroid estrone. These molecules were equipped with cationic residues yielding amphiphiles suitable for lipofection. To this end, self-assembled luminescent structures were formed in aqueous media and mixed with a red-fluorescent protein expressing plasmid, yielding lipoplexes with increased emission intensity. These luminescent lipoplexes were able to efficiently transfect HeLa and HEK 293T cells and were able to be tracked due to the aggregation induced-emission properties.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures
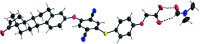

Similar articles
-
Luminescent Amphiphilic Aminoglycoside Probes to Study Transfection.Chembiochem. 2021 May 4;22(9):1563-1567. doi: 10.1002/cbic.202000725. Epub 2021 Feb 11. Chembiochem. 2021. PMID: 33410196 Free PMC article.
-
Deoxyestrone-based lipofection agents with solution- and solid-state emission properties.Org Biomol Chem. 2023 Jul 19;21(28):5762-5767. doi: 10.1039/d3ob00656e. Org Biomol Chem. 2023. PMID: 37404013
-
Recent Advances in Aggregation-Induced Electrochemiluminescence.Chemistry. 2019 Oct 1;25(55):12671-12683. doi: 10.1002/chem.201902465. Epub 2019 Aug 8. Chemistry. 2019. PMID: 31283848 Review.
-
Achieving very bright mechanoluminescence from purely organic luminophores with aggregation-induced emission by crystal design.Chem Sci. 2016 Aug 1;7(8):5307-5312. doi: 10.1039/c6sc01325b. Epub 2016 Apr 26. Chem Sci. 2016. PMID: 30155182 Free PMC article.
-
Luminescent Organometallic Nanomaterials with Aggregation-Induced Emission.Crit Rev Anal Chem. 2018 Jul 4;48(4):330-336. doi: 10.1080/10408347.2018.1445960. Epub 2018 Mar 20. Crit Rev Anal Chem. 2018. PMID: 29557670 Review.
Cited by
-
Advances towards Cell-Specific Gene Transfection: A Small-Molecule Approach Allows Order-of-Magnitude Selectivity.Chemistry. 2022 Aug 1;28(43):e202104618. doi: 10.1002/chem.202104618. Epub 2022 Jun 17. Chemistry. 2022. PMID: 35604769 Free PMC article.
-
Luminescent Amphiphilic Aminoglycoside Probes to Study Transfection.Chembiochem. 2021 May 4;22(9):1563-1567. doi: 10.1002/cbic.202000725. Epub 2021 Feb 11. Chembiochem. 2021. PMID: 33410196 Free PMC article.
References
-
- LeWitt P. A. Rezai A. R. Leehey M. A. Ojemann S. G. Flaherty A. W. Eskandar E. N. Kostyk S. K. Thomas K. Sarkar A. Siddiqui M. S. Tatter S. B. Schwalb J. M. Poston K. L. Henderson J. M. Kurlan R. M. Richard I. H. Van Meter L. Sapan C. V. During M. J. Kaplitt M. G. Feigin A. Lancet Neurol. 2011;10:309–319. doi: 10.1016/S1474-4422(11)70039-4. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

