The effect of PEG functionalization on the in vivo behavior and toxicity of CdTe quantum dots
- PMID: 35515837
- PMCID: PMC9063558
- DOI: 10.1039/c9ra00022d
The effect of PEG functionalization on the in vivo behavior and toxicity of CdTe quantum dots
Abstract
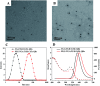
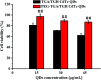
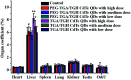
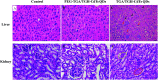
CdTe quantum dots (QDs) are considered a potential toxic substance because they contain metal ions. However, most toxicology data are derived from in vitro studies or limited in vivo analysis and may not reflect in vivo responses and biodistribution. Proper modification is one of the most widely used routes to reduce the toxicity of QDs. Herein, we demonstrated the role of polyethylene glycol (PEG) in decreasing the toxicity of QDs by studying the animal survival, clinical biochemistry, organ histology, biodistribution and oxidative stress in thioglycolic acid (TGA)- and mercapto-acetohydrazide (TGH)-stabilized CdTe QD (TGA/TGH-CdTe QD)-treated groups. Via the histology, transmission electron microscopy (TEM) and biodistribution results, it was found that the QDs mainly accumulated in the liver and kidney at 7 days post-injection, and obvious tissue damage was also observed in the bare TGA/TGH-CdTe QD group. Based on the evaluation of oxidative stress in the liver and kidney, the indicators exhibited an obvious variation with a high dose of TGA/TGH-CdTe QDs. In contrast, the QD aggregation decreased in the liver and kidney with no clear physiological index variation after PEG functionalization. Thus, PEG plays an important role in decreasing the toxicity of the CdTe QDs, and both the accumulation of cadmium and oxidative stress variation instead of an isolation factor are responsible for the in vivo toxicity of these QDs.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures

Similar articles
-
Preparation of pH-stimuli-responsive PEG-TGA/TGH-capped CdTe QDs and their application in cell labeling.Luminescence. 2015 Aug;30(5):519-25. doi: 10.1002/bio.2770. Epub 2014 Sep 22. Luminescence. 2015. PMID: 25244429
-
Influence of different functionalized CdTe quantum dots on the accumulation of metals, developmental toxicity and respiration in different development stages of the zebrafish (Danio rerio).Front Toxicol. 2023 May 2;5:1176172. doi: 10.3389/ftox.2023.1176172. eCollection 2023. Front Toxicol. 2023. PMID: 37200940 Free PMC article.
-
Thermodynamic and kinetic insights into the interactions between functionalized CdTe quantum dots and human serum albumin: A surface plasmon resonance approach.Int J Biol Macromol. 2021 Aug 1;184:990-999. doi: 10.1016/j.ijbiomac.2021.06.158. Epub 2021 Jun 28. Int J Biol Macromol. 2021. PMID: 34197852
-
The cytotoxicity of cadmium-based quantum dots.Biomaterials. 2012 Feb;33(5):1238-44. doi: 10.1016/j.biomaterials.2011.10.070. Epub 2011 Nov 10. Biomaterials. 2012. PMID: 22078811 Review.
-
Population pharmacokinetic modelling of indium-based quantum dot nanoparticles: preclinical in vivo studies.Eur J Pharm Sci. 2021 Feb 1;157:105639. doi: 10.1016/j.ejps.2020.105639. Epub 2020 Nov 11. Eur J Pharm Sci. 2021. PMID: 33188925 Free PMC article. Review.
Cited by
-
Enhancing the solubility and potency of tetrahydrocurcumin as an anti-cancer agent using a β-cyclodextrin inclusion complex approach.PLoS One. 2024 Jul 26;19(7):e0305171. doi: 10.1371/journal.pone.0305171. eCollection 2024. PLoS One. 2024. PMID: 39058699 Free PMC article.
-
Revisiting the cytotoxicity of quantum dots: an in-depth overview.Biophys Rev. 2020 Jun;12(3):703-718. doi: 10.1007/s12551-020-00653-0. Epub 2020 Mar 5. Biophys Rev. 2020. PMID: 32140918 Free PMC article. Review.
-
Advancing Cancer Therapy with Quantum Dots and Other Nanostructures: A Review of Drug Delivery Innovations, Applications, and Challenges.Cancers (Basel). 2025 Mar 4;17(5):878. doi: 10.3390/cancers17050878. Cancers (Basel). 2025. PMID: 40075725 Free PMC article. Review.
-
Designing the Surface Chemistry of Inorganic Nanocrystals for Cancer Imaging and Therapy.Cancers (Basel). 2022 May 16;14(10):2456. doi: 10.3390/cancers14102456. Cancers (Basel). 2022. PMID: 35626059 Free PMC article. Review.
-
About the Influence of PEG Spacers on the Cytotoxicity of Titanate Nanotubes-Docetaxel Nanohybrids against a Prostate Cancer Cell Line.Nanomaterials (Basel). 2021 Oct 15;11(10):2733. doi: 10.3390/nano11102733. Nanomaterials (Basel). 2021. PMID: 34685172 Free PMC article.
References
LinkOut - more resources
Full Text Sources

