Product distribution and mechanism of the OH- initiated tropospheric degradation of three CFC replacement candidates: CH3CF[double bond, length as m-dash]CH2, (CF3)2C[double bond, length as m-dash]CH2 and (E/ Z)-CF3CF[double bond, length as m-dash]CHF
- PMID: 35515909
- PMCID: PMC9060772
- DOI: 10.1039/c8ra09627a
Product distribution and mechanism of the OH- initiated tropospheric degradation of three CFC replacement candidates: CH3CF[double bond, length as m-dash]CH2, (CF3)2C[double bond, length as m-dash]CH2 and (E/ Z)-CF3CF[double bond, length as m-dash]CHF
Abstract
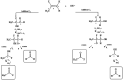
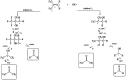
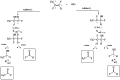
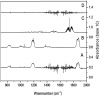
The OH radical initiated photodegradation of 2-fluoropropene (CH3CF[double bond, length as m-dash]CH2), 3,3,3-trifluoro-2-(tri-fluoromethyl)propene ((CF3)2C[double bond, length as m-dash]CH2) and (E/Z)-1,2,3,3,3-pentafluoropropene ((E/Z)-CF3CF[double bond, length as m-dash]CHF) has been investigated for the first time using a 1080 L quartz-glass environmental chamber at 298 ± 2 K and atmospheric pressure of synthetic air coupled with in situ FTIR spectroscopy to monitor reactants and products. The major products observed in the OH reaction were CH3C(O)F (98 ± 5)% together with HC(O)H (89 ± 7)% as a co-product, CF3C(O)F (103 ± 8)% together with HC(O)F (96 ± 7)% as a co-product and CF3C(O)CF3 (91 ± 8)% together with HC(O)H (98 ± 12)% as a co-product from the C1-C2 bond cleavage channel of the intermediate hydroxyalkoxy radical, formed by addition of OH to the terminal carbon of the double bond which is designated C1 of 2-fluoropropene, (E/Z)-1,2,3,3,3-pentafluoropropene and 3,3,3-trifluoro-2-(tri-fluoromethyl)propene, respectively. The present results are compared with previous studies for the reaction of OH with the separate isomers (E) and (Z) of 1,2,3,3,3-pentafluoropropene. In addition, atmospheric implications of the reactions studied are discussed.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures

References
-
- Prather M. J. Hsu J. NF3, the greenhouse gas missing from Kyoto. Geophys. Res. Lett. 2008;35(12):L12810. doi: 10.1029/2008GL034542. - DOI
-
- Montzka S. A. McFarland M. Andersen S. O. Miller B. R. Fahey D. W. Hall B. D. Hu L. Siso C. Elkins J. W. Recent Trends in Global Emissions of Hydrochlorofluorocarbons and Hydrofluorocarbons: Reflecting on the 2007 Adjustments to the Montreal Protocol. J. Phys. Chem. A. 2015;119(19):4439–4449. doi: 10.1021/jp5097376. - DOI - PubMed
-
- U.S. Environmental Protection Agency, U.S. EPA: Motor Vehicle Air Conditioners, http://opusinspection.com/documents/
-
- Reisch M. S. New refrigerant takes heat. Chem. Eng. News. 2008;86(46):35–36. doi: 10.1021/cen-v086n046.p035. - DOI
LinkOut - more resources
Full Text Sources

