Bismuth subsalicylate, a low-toxicity catalyst for the ring-opening polymerization (ROP) of l-lactide (l-LA) with aliphatic diol initiators: synthesis, characterization, and mechanism of initiation
- PMID: 35516064
- PMCID: PMC9057703
- DOI: 10.1039/d0ra05413e
Bismuth subsalicylate, a low-toxicity catalyst for the ring-opening polymerization (ROP) of l-lactide (l-LA) with aliphatic diol initiators: synthesis, characterization, and mechanism of initiation
Abstract
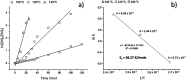
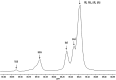
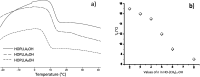
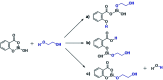
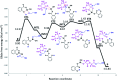
The ring-opening polymerization (ROP) of l-lactide (l-LA) was induced by the catalytic action of bismuth subsalicylate (BiSS) using linear aliphatic diols [HO(CH2) n OH, where n = 2, 3, 4, 5, 6, and 8] as initiators and chain transfer agents. The theoretical and experimental degree of polymerization (DP) in all samples of α,ω-hydroxy telechelic poly(l-lactide) (HOPLLAOH) had a good agreement in all samples, an effect attributed to the interaction of BiSS with HO(CH2) n OH inducing a transfer reaction. HOPLLAOH was synthesized and characterized by a range of analytical techniques, confirming the insertion of methylene groups from the initiator into the main chain of the polyester. The glass-transition temperature (T g) of HOPLLAOH was found to be proportional to the number of methylene groups present in the diol. Various parameters regarding the ROP of l-LA were studied, such as temperature, time of reaction, amount of catalyst, and the nature of the diols. A kinetic study of the reaction allowed the determination of the rate constants (k) and activation energy (E a). A mechanism of initiation is proposed based on a computational study using density functional theory (DFT), evidencing the role of the alkyl diol as an initiator, producing an alkoxide (Bi-OROH). This species then acts as a nucleophile, attacking the carbonyl group, inducing its insertion, and ultimately completing the ring-opening of l-LA.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures
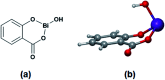
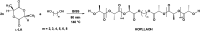



Similar articles
-
Synthesis and characterization of macrodiols and non-segmented poly(ester-urethanes) (PEUs) derived from α,ω-hydroxy telechelic poly(ε-caprolactone) (HOPCLOH): effect of initiator, degree of polymerization, and diisocyanate.RSC Adv. 2024 Aug 27;14(37):27241-27251. doi: 10.1039/d4ra03951c. eCollection 2024 Aug 22. RSC Adv. 2024. PMID: 39193304 Free PMC article.
-
A Comprehensive Investigation of the Structural, Thermal, and Biological Properties of Fully Randomized Biomedical Polyesters Synthesized with a Nontoxic Bismuth(III) Catalyst.Molecules. 2022 Feb 8;27(3):1139. doi: 10.3390/molecules27031139. Molecules. 2022. PMID: 35164403 Free PMC article.
-
Highly Effective and Green Catalytic Approach Toward α,ω-Dihydroxy-Telechelic Poly(trimethylenecarbonate).Macromol Rapid Commun. 2009 Dec 16;30(24):2128-35. doi: 10.1002/marc.200900498. Epub 2009 Sep 3. Macromol Rapid Commun. 2009. PMID: 21638506
-
Indium Catalysts for Ring Opening Polymerization: Exploring the Importance of Catalyst Aggregation.Acc Chem Res. 2017 Nov 21;50(11):2861-2869. doi: 10.1021/acs.accounts.7b00447. Epub 2017 Oct 31. Acc Chem Res. 2017. PMID: 29087695 Review.
-
DFT Modeling of Organocatalytic Ring-Opening Polymerization of Cyclic Esters: A Crucial Role of Proton Exchange and Hydrogen Bonding.Polymers (Basel). 2019 Dec 12;11(12):2078. doi: 10.3390/polym11122078. Polymers (Basel). 2019. PMID: 31842423 Free PMC article. Review.
Cited by
-
Monodisperse oligo(ε-caprolactones) with terpenes and alkyl end-groups: synthesis, isolation, characterization, and antibacterial activity.RSC Adv. 2025 Jan 2;15(1):276-288. doi: 10.1039/d4ra08104h. eCollection 2025 Jan 2. RSC Adv. 2025. PMID: 39758912 Free PMC article.
-
Comparison of three elements (In, Sn, and Sb) in the same period as catalysts in the ring-opening polymerization of l-lactide: from amorphous to semicrystalline polyesters.RSC Adv. 2024 Oct 31;14(47):34733-34745. doi: 10.1039/d4ra06783e. eCollection 2024 Oct 29. RSC Adv. 2024. PMID: 39483385 Free PMC article.
-
Bioactive Bismuth Compounds: Is Their Toxicity a Barrier to Therapeutic Use?Int J Mol Sci. 2024 Jan 27;25(3):1600. doi: 10.3390/ijms25031600. Int J Mol Sci. 2024. PMID: 38338879 Free PMC article. Review.
-
A Highly Active and Selective Zirconium-Based Catalyst System for the Industrial Production of Poly(lactic acid).ACS Catal. 2023 Feb 7;13(4):2681-2695. doi: 10.1021/acscatal.2c05690. eCollection 2023 Feb 17. ACS Catal. 2023. PMID: 36846823 Free PMC article.
-
Poly lactic acid production using the ring opening polymerization (ROP) method using Lewis acid surfactant combined iron (Fe) catalyst (Fe(DS)3).Heliyon. 2023 Jul 7;9(8):e17985. doi: 10.1016/j.heliyon.2023.e17985. eCollection 2023 Aug. Heliyon. 2023. PMID: 37520956 Free PMC article.
References
-
- Ji H. Wang B. Pan L. Li Y. S. Green Chem. 2018;20:641–648. doi: 10.1039/C7GC03261G. - DOI
-
- Ji H. Chen X. Wang B. Pan L. Li Y. S. Green Chem. 2018;20:3963–3973. doi: 10.1039/C8GC01641K. - DOI
-
- Shi T. Zheng Q. Zuo W. Liu S. Li Z. Chin. J. Polym. Sci. 2018;36:149–156. doi: 10.1007/s10118-018-2039-5. - DOI
LinkOut - more resources
Full Text Sources

