Non-aqueous solvent extraction of indium from an ethylene glycol feed solution by the ionic liquid Cyphos IL 101: speciation study and continuous counter-current process in mixer-settlers
- PMID: 35516195
- PMCID: PMC9055152
- DOI: 10.1039/d0ra04684a
Non-aqueous solvent extraction of indium from an ethylene glycol feed solution by the ionic liquid Cyphos IL 101: speciation study and continuous counter-current process in mixer-settlers
Abstract
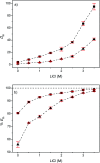
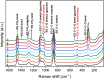
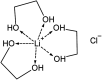
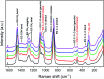
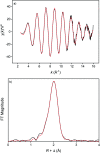
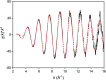
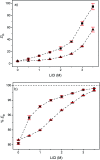
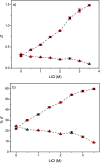
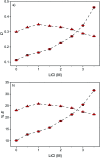
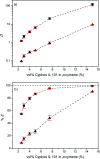
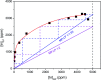
A solvometallurgical process for the separation of indium(iii) and zinc(ii) from ethylene glycol solutions using the ionic liquid extractants Cyphos IL 101 and Aliquat 336 in an aromatic diluent has been investigated. The speciation of indium(iii) in the two immiscible organic phases was investigated by Raman spectroscopy, infrared spectroscopy, EXAFS and 115In NMR spectroscopy. At low LiCl concentrations in ethylene glycol, the bridging (InCl3)2(EG)3 or mononuclear (InCl3)(EG)2 complex is proposed. At higher lithium chloride concentrations, the first coordination sphere changes to two oxygen atoms from one bidentate ethylene glycol ligand and four chloride anions ([In(EG)Cl4]-). In the less polar phase, indium(iii) is present as a tetrahedral [InCl4]- complex independent of the LiCl concentration. After the number of theoretical stages had been determined using a McCabe-Thiele diagram for extraction by Cyphos IL 101, the extraction and scrubbing processes were performed in lab-scale mixer-settlers to test the feasibility of working in continuous mode. Indium(iii) was extracted quantitatively in four stages, with 19% co-extraction of zinc(ii). The co-extracted zinc(ii) was scrubbed selectively in six stages using an indium(iii) scrub solution. Indium(iii) was recovered from the loaded less polar organic phase as indium(iii) hydroxide (98.5%) by precipitation stripping with an aqueous NaOH solution.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures
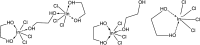

Similar articles
-
Speciation of indium(iii) chloro complexes in the solvent extraction process from chloride aqueous solutions to ionic liquids.Dalton Trans. 2017 Mar 27;46(13):4412-4421. doi: 10.1039/c7dt00618g. Dalton Trans. 2017. PMID: 28294262
-
Separation of iron(iii), zinc(ii) and lead(ii) from a choline chloride-ethylene glycol deep eutectic solvent by solvent extraction.RSC Adv. 2020 Sep 8;10(55):33161-33170. doi: 10.1039/d0ra06091g. eCollection 2020 Sep 7. RSC Adv. 2020. PMID: 35515064 Free PMC article.
-
Separation of neodymium and dysprosium by solvent extraction using ionic liquids combined with neutral extractants: batch and mixer-settler experiments.RSC Adv. 2020 Jan 2;10(1):307-316. doi: 10.1039/c9ra08996a. eCollection 2019 Dec 20. RSC Adv. 2020. PMID: 35492521 Free PMC article.
-
Recovery of pure germanium oxide from Zener diodes using a recyclable ionic liquid Cyphos IL 104.J Environ Manage. 2020 Dec 15;276:111218. doi: 10.1016/j.jenvman.2020.111218. Epub 2020 Sep 1. J Environ Manage. 2020. PMID: 32882518
-
Phosphonium ionic liquids as extractants for recovery of ruthenium(III) from acidic aqueous solutions.Chem Zvesti. 2017;71(6):1065-1072. doi: 10.1007/s11696-016-0027-1. Epub 2016 Dec 21. Chem Zvesti. 2017. PMID: 28553005 Free PMC article.
Cited by
-
Applied novel functionality in separation procedure from leaching solution of zinc plant residue by using non-aqueous solvent extraction.Sci Rep. 2023 Jan 20;13(1):1146. doi: 10.1038/s41598-023-27646-9. Sci Rep. 2023. PMID: 36670143 Free PMC article.
-
Liquid-Liquid Extraction of Indium(III) from the HCl Medium by Ionic Liquid A327H+Cl- and Its Use in a Supported Liquid Membrane System.Molecules. 2020 Nov 10;25(22):5238. doi: 10.3390/molecules25225238. Molecules. 2020. PMID: 33182748 Free PMC article.
-
Nonaqueous Solvent Extraction for Enhanced Metal Separations: Concept, Systems, and Mechanisms.Ind Eng Chem Res. 2021 Dec 8;60(48):17285-17302. doi: 10.1021/acs.iecr.1c02287. Epub 2021 Nov 19. Ind Eng Chem Res. 2021. PMID: 34898845 Free PMC article. Review.
-
Computational and Experimental Investigation of the Selective Adsorption of Indium/Iron Ions by the Epigallocatechin Gallate Monomer.Materials (Basel). 2022 Nov 21;15(22):8251. doi: 10.3390/ma15228251. Materials (Basel). 2022. PMID: 36431735 Free PMC article.
-
Assessment of metal extraction from e-waste using supported IL membrane with reliable comparison between RSM regression and ANN framework.Sci Rep. 2024 Feb 16;14(1):3882. doi: 10.1038/s41598-024-54591-y. Sci Rep. 2024. PMID: 38366075 Free PMC article.
References
-
- Pradhan D. Panda S. Sukla L. B. Miner. Process. Extr. Metall. Rev. 2018;39:167–180. doi: 10.1080/08827508.2017.1399887. - DOI
-
- Alfantazi A. M. Moskalyk R. R. Miner. Eng. 2003;16:687–694. doi: 10.1016/S0892-6875(03)00168-7. - DOI
-
- Felix N., in Ullmann's Encyclopedia of Industrial Chemistry, ed. F. Ullmann, Wiley-VCH, Weinheim, 2012, pp. 65–74
-
- Nguyen T. H. Man Seung L. Miner. Process. Extr. Metall. Rev. 2019;40:278–291. doi: 10.1080/08827508.2018.1538987. - DOI
-
- Paiva A. P. Sep. Sci. Technol. 2001;36:1395–1419. doi: 10.1081/SS-100103878. - DOI
LinkOut - more resources
Full Text Sources
Miscellaneous

