Preparation of quercetin-nicotinamide cocrystals and their evaluation under in vivo and in vitro conditions
- PMID: 35516602
- PMCID: PMC9054514
- DOI: 10.1039/d0ra03324c
Preparation of quercetin-nicotinamide cocrystals and their evaluation under in vivo and in vitro conditions
Abstract
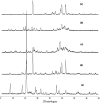
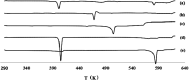
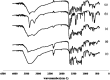
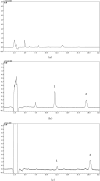
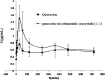
Quercetin is a flavonoid abundant in the plant kingdom. Various types of bioactivities of quercetin have been demonstrated in vitro. Although quercetin has been proposed to exhibit numerous pharmacological benefits, it suffers from low bioavailability on account of its obviously poor solubility in water. Cocrystals have generated interest recently as a way of enhancing the dissolution in vitro and creating relative bioavailability of insoluble medicine. In this study, quercetin-nicotinamide cocrystals were obtained via a solvent evaporation technique. Furthermore, quercetin-nicotinamide cocrystals were characterized via Fourier transform infrared (FI-IR) spectroscopy, X-ray powder diffraction (PXRD), and differential scanning calorimetry (DSC) techniques. Quercetin-nicotinamide cocrystals are a new phase material, and the established intermolecular forces such as hydrogen bonds between quercetin and nicotinamide existed in the quercetin-nicotinamide cocrystals, as confirmed from the solid-state analysis. The dissolution of quercetin-nicotinamide cocrystals was found to be significantly higher than that of quercetin crystals. The pharmacokinetic data from the in vivo experiments suggested that quercetin-nicotinamide cocrystals could significantly increase the oral absorption of quercetin by nearly 4-fold. These results demonstrate that the developed quercetin-nicotinamide cocrystals are a promising oral formulation toward improvement in the dissolution and bioavailability of quercetin.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures

Similar articles
-
Cocrystals of quercetin with improved solubility and oral bioavailability.Mol Pharm. 2011 Oct 3;8(5):1867-76. doi: 10.1021/mp200209j. Epub 2011 Aug 25. Mol Pharm. 2011. PMID: 21846121
-
Chrysin cocrystals: Characterization and evaluation.J Pharm Biomed Anal. 2017 Feb 5;134:361-371. doi: 10.1016/j.jpba.2016.10.020. Epub 2016 Nov 3. J Pharm Biomed Anal. 2017. PMID: 27894779
-
Effect of stoichiometry upon the characteristics of quercetin-arginine cocrystals formulated through solution crystallization.Drug Dev Ind Pharm. 2024 Feb;50(2):163-172. doi: 10.1080/03639045.2024.2306281. Epub 2024 Jan 24. Drug Dev Ind Pharm. 2024. PMID: 38226968
-
Improvement strategies for the oral bioavailability of poorly water-soluble flavonoids: An overview.Int J Pharm. 2019 Oct 30;570:118642. doi: 10.1016/j.ijpharm.2019.118642. Epub 2019 Aug 22. Int J Pharm. 2019. PMID: 31446024 Review.
-
Recent advances in improving oral drug bioavailability by cocrystals.Bioimpacts. 2018;8(4):305-320. doi: 10.15171/bi.2018.33. Epub 2018 May 31. Bioimpacts. 2018. PMID: 30397585 Free PMC article. Review.
Cited by
-
A Systematic Review: Quercetin-Secondary Metabolite of the Flavonol Class, with Multiple Health Benefits and Low Bioavailability.Int J Mol Sci. 2024 Nov 11;25(22):12091. doi: 10.3390/ijms252212091. Int J Mol Sci. 2024. PMID: 39596162 Free PMC article.
-
Preparation of caffeic acid grafted chitosan self-assembled micelles to enhance oral bioavailability and antibacterial activity of quercetin.Front Vet Sci. 2023 Jul 5;10:1218025. doi: 10.3389/fvets.2023.1218025. eCollection 2023. Front Vet Sci. 2023. PMID: 37476826 Free PMC article.
-
Flavonoids from Clerodendrum genus and their biological activities.ADMET DMPK. 2024 Dec 10;12(6):843-879. doi: 10.5599/admet.2442. eCollection 2024. ADMET DMPK. 2024. PMID: 39713250 Free PMC article. Review.
-
Potential of Quercetin as a Promising Therapeutic Agent Against Type 2 Diabetes.Molecules. 2025 Jul 24;30(15):3096. doi: 10.3390/molecules30153096. Molecules. 2025. PMID: 40807271 Free PMC article. Review.
-
Targeting Senescence, Oxidative Stress, and Inflammation: Quercetin-Based Strategies for Ocular Diseases in Older Adults.Clin Interv Aging. 2025 Jun 7;20:791-813. doi: 10.2147/CIA.S516946. eCollection 2025. Clin Interv Aging. 2025. PMID: 40503074 Free PMC article. Review.
References
-
- Hoensch H. P. Oertel R. The value of flavonoids for the human nutrition: short review and perspectives. Clin. Nutr. Exp. 2015;3:8–14. doi: 10.1016/j.yclnex.2015.09.001. - DOI
-
- Aune D. Giovannucci E. Boffetta P. Fadnes L. T. Keum N. Norat T. et al., Fruit and vegetable intake and the risk of cardiovascular disease, total cancer and all-cause mortality-a systematic review and dose-response meta-analysis of prospective studies. Int. J. Epidemiol. 2017;46(3):1029–1056. doi: 10.1093/ije/dyw319. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

