Resonance-assisted/impaired anion-π interaction: towards the design of novel anion receptors
- PMID: 35517107
- PMCID: PMC9056982
- DOI: 10.1039/d0ra07877h
Resonance-assisted/impaired anion-π interaction: towards the design of novel anion receptors
Abstract
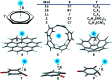
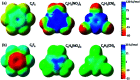
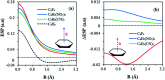
Substituents alter the electron density distribution in benzene in various ways, depending on their electron withdrawing and donating capabilities, as summarized by the empirical Hammett equation. The change of the π electron density distribution subsequently impacts the interaction of substituted benzenes or other cyclic conjugated rings with anions. Currently the design and synthesis of conjugated cyclic receptors capable of binding anions is an active field due to their applications in the sensing and removal of environmental contaminants and molecular recognition. By using the block-localized wavefunction (BLW) method, which is a variant of ab initio valence bond (VB) theory and can derive the reference resonance-free state self-consistently, we quantified the resonance-assisted (RA) or resonance-impaired (RI) phenomena in anion-π interactions from both structural and energetic perspectives. The frozen interaction, in which the electrostatic attraction is involved, has been shown to be the governing factor for the RA or RI interactions with anions. Energy analyses based on the empirical point charge (EPC) model indicated that the anion-π interactions can be simplified as the attraction between a negative point charge (anion) and a group of local dipoles, affected by the enriched or diminished π-cloud due to the resonance between the substituents and the conjugated ring. Hence, two strategies for the design of novel anion receptors can be envisioned. One is the enhancement of the magnitudes and/or numbers of local dipoles (polarized σ bonds), and the other is the reduction of π electron density in conjugated rings. For cases with the RI characteristics, "curved" aromatic molecules are preferred to be anion receptors. Indeed, extremely strong binding was found in complexes formed with fluorinated corannulene (F-CDD) and fluorinated [5]cycloparaphenylene (F-[5]CPP). Inspired by the RA phenomenon, complexes of p-, o- and m-benzoquinones with halides were revisited.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures
Similar articles
-
Are anion/pi interactions actually a case of simple charge-dipole interactions?J Phys Chem A. 2010 Aug 26;114(33):8658-64. doi: 10.1021/jp1010549. J Phys Chem A. 2010. PMID: 20433187 Free PMC article.
-
The strength and selectivity of perfluorinated nano-hoops and buckybowls for anion binding and the nature of anion-π interactions.J Comput Chem. 2023 Jan 30;44(3):138-148. doi: 10.1002/jcc.26820. Epub 2022 Feb 11. J Comput Chem. 2023. PMID: 35147229
-
Predicting the Strength of Anion-π Interactions of Substituted Benzenes: the Development of Anion-π Binding Substituent Constants.J Phys Chem A. 2016 Nov 23;120(46):9235-9243. doi: 10.1021/acs.jpca.6b06276. Epub 2016 Nov 9. J Phys Chem A. 2016. PMID: 27779403
-
Ion-π interactions in ligand design for anions and main group cations.Acc Chem Res. 2013 Apr 16;46(4):955-66. doi: 10.1021/ar300100g. Epub 2012 Jun 22. Acc Chem Res. 2013. PMID: 22726207 Free PMC article.
-
Anion-Induced Electron Transfer.Acc Chem Res. 2018 Sep 18;51(9):2225-2236. doi: 10.1021/acs.accounts.8b00197. Epub 2018 Sep 7. Acc Chem Res. 2018. PMID: 30192503
Cited by
-
An Investigation into Anion Sensing of the Molecular Aggregate of 4-(Pyrrol-1-yl)pyridine and Its Derivatives.Molecules. 2024 Dec 2;29(23):5692. doi: 10.3390/molecules29235692. Molecules. 2024. PMID: 39683851 Free PMC article.
References
-
- Hobza P. and Müller-Dethlefs K., Non-covalent interactions: theory and experiment, Royal Society of Chemistry, 2010
-
- Lee J. Y. Hong B. H. Kim W. Y. Min S. K. Kim Y. Jouravlev M. V. Bose R. Kim K. S. Hwang I.-C. Kaufman L. J. Wong C. W. Kim P. Kim K. S. Nature. 2009;460:498–501. doi: 10.1038/nature08173. - DOI
-
- Salonen L. M. Ellermann M. Diederich F. Angew. Chem., Int. Ed. 2011;50:4808–4842. doi: 10.1002/anie.201007560. - DOI
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

