Electrochemical synthesis and corrosion protection of poly(3-aminophenylboronic acid- co-pyrrole) on mild steel
- PMID: 35517519
- PMCID: PMC9057268
- DOI: 10.1039/d0ra07311c
Electrochemical synthesis and corrosion protection of poly(3-aminophenylboronic acid- co-pyrrole) on mild steel
Abstract
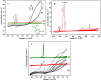
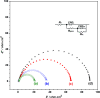
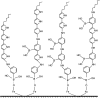
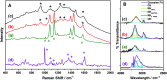
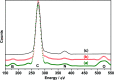
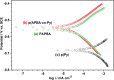
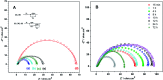
Synthesis of poly(3-aminophenylboronic acid-co-pyrrole) (p(APBA-co-Py)) is carried out potentiodynamically on a pre-passivated mild steel (MS) surface in an oxalic acid solution containing 3-aminophenylboronic acid (APBA) and pyrrole (Py) monomers. The monomer feed ratio was determined using electrochemical impedance spectroscopy (EIS) and adhesion tests. The p(APBA-co-Py) coating is characterized by electrochemically and spectroscopically comparing with poly(3-aminophenylboronic acid) (p(APBA) and polypyrrole (p(Py) homopolymers. SERS, FTIR, XPS, scanning electron microscopy-wavelength dispersive X-ray and- energy dispersive X-ray spectroscopy results indicate the presence of both APBA and Py segments in the p(APBA-co-Py) backbone. The protective properties of the coating are investigated by Tafel and EIS measurements in a 0.50 M HCl solution. The corrosion resistance of p(APBA-co-Py)-coated MS (66.8 Ω cm2) is higher than that of p(APBA)- and p(Py)-coated, passivated, and uncoated MS. The p(APBA-co-Py) coating embodies the advantageous features of both homopolymers. Py units in p(APBA-co-Py) chains improve the protective property while APBA units carrying the -B(OH)2 group develop the adhesive property of the layer. EIS results show that the p(APBA-co-Py) coating, due to its homogeneous and compact distribution and the formation of a stable interface, enhanced corrosion resistance of MS by 87.4% for 10 hours in HCl corrosive medium.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures


Similar articles
-
Characterization of PTFE Film on 316L Stainless Steel Deposited through Spin Coating and Its Anticorrosion Performance in Multi Acidic Mediums.Materials (Basel). 2020 Jan 14;13(2):388. doi: 10.3390/ma13020388. Materials (Basel). 2020. PMID: 31947700 Free PMC article.
-
Protective layer formation on magnesium in cell culture medium.Mater Sci Eng C Mater Biol Appl. 2016 Jun;63:341-51. doi: 10.1016/j.msec.2016.03.003. Epub 2016 Mar 3. Mater Sci Eng C Mater Biol Appl. 2016. PMID: 27040228
-
Poly(pyrrole-co-o-toluidine) wrapped CoFe2O4/R(GO-OXSWCNTs) ternary composite material for Ga3+ sensing ability.RSC Adv. 2019 Oct 16;9(57):33052-33070. doi: 10.1039/c9ra03593a. eCollection 2019 Oct 15. RSC Adv. 2019. PMID: 35529122 Free PMC article.
-
Exploration of Sunflower Oil As a Renewable Biomass Source to Develop Scalable and Highly Effective Corrosion Inhibitors in a 15% HCl Medium at High Temperatures.ACS Appl Mater Interfaces. 2021 Jan 20;13(2):3119-3138. doi: 10.1021/acsami.0c18887. Epub 2021 Jan 11. ACS Appl Mater Interfaces. 2021. PMID: 33423454
-
Electrochemically Produced Copolymers of Pyrrole and Its Derivatives: A Plentitude of Material Properties Using "Simple" Heterocyclic Co-Monomers.Materials (Basel). 2021 Jan 7;14(2):281. doi: 10.3390/ma14020281. Materials (Basel). 2021. PMID: 33430477 Free PMC article. Review.
References
-
- Schweitzer P. A., Fundamentals of Corrosion, CRC Press, USA, 2010
-
- Baboıan R., Corrosion Tests and Standards, ed. R. Baboian, W. Sheldon. J. Dean, H. P. Hack, E. L. Hibner and J. R. Scully, ASTM INTERNATIONAL, USA, 2 edn, 2010, pp. 1–882
-
- Revie R. W. and Uhlig H. H., Corrosion and Corrosion Control, Wiley-Interscience, New Jersey, 4 edn, 2008, pp. 1–490
-
- Su C. Ji L. Xu L. Zhu X. He H. Lv Y. et al., A novel ferrocene-containing aniline copolymer: its synthesis and electrochemical performance. RSC Adv. 2015;5(18):14053–14060. doi: 10.1039/C4RA12476F. - DOI
-
- Olgun U. Gülfen M. Effects of different dopants on the band gap and electrical conductivity of the poly(phenylene-thiazolo[5,4-d]thiazole) copolymer. RSC Adv. 2014;4(48):25165–25171. doi: 10.1039/C4RA02425G. - DOI
LinkOut - more resources
Full Text Sources
Miscellaneous

