Unusual features of nitrogen substitutions in silicene
- PMID: 35518139
- PMCID: PMC9056544
- DOI: 10.1039/d0ra04470a
Unusual features of nitrogen substitutions in silicene
Abstract
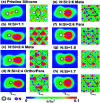
The quasiparticle properties resulting from charge and spin are clearly identified in nitrogen-substituted silicenes, for which a theoretical framework is successfully developed from first-principles calculations. Such systems create extremely non-uniform chemical and physical environments through the distribution of the guest atoms. They present unusual geometric, electronic, and magnetic properties, which can be identified from the optimal honeycomb lattices, the atom- and spin-dominated energy spectra, the spatial charge density distributions, and the atom-, orbital- and spin-projected van Hove singularities [the net magnetic moments]. The complicated relations between the highly hybridized sp2-N-Si bonds and the ferromagnetic/non-magnetic configurations are responsible for the p-type or semiconducting behavior, the significant modifications to the Dirac cone structures, the difficulty in identifying the π and σ bands, and the vanishing or finite magnetic moments. The theoretical predictions could be verified by high-resolution experimental measurements.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures




Similar articles
-
Rich p-type-doping phenomena in boron-substituted silicene systems.R Soc Open Sci. 2020 Dec 2;7(12):200723. doi: 10.1098/rsos.200723. eCollection 2020 Dec. R Soc Open Sci. 2020. PMID: 33489254 Free PMC article.
-
Stacking-configuration-enriched essential properties of bilayer graphenes and silicenes.J Chem Phys. 2020 Oct 21;153(15):154707. doi: 10.1063/5.0024421. J Chem Phys. 2020. PMID: 33092355
-
Fundamental Properties of Transition-Metals-Adsorbed Graphene.Chemphyschem. 2019 Oct 2;20(19):2473-2481. doi: 10.1002/cphc.201900614. Epub 2019 Aug 27. Chemphyschem. 2019. PMID: 31353787
-
Muonium as a model for interstitial hydrogen in the semiconducting and semimetallic elements.Rep Prog Phys. 2009 Oct 15;72(11). doi: 10.1088/0034-4885/72/11/116501. Rep Prog Phys. 2009. PMID: 35172524 Review.
-
Moiré-Potential-Induced Band Structure Engineering in Graphene and Silicene.Small. 2021 Mar;17(9):e1903769. doi: 10.1002/smll.201903769. Epub 2019 Sep 18. Small. 2021. PMID: 31531941 Review.
Cited by
-
Tunable Electronic Properties of Two-Dimensional GaSe1-xTex Alloys.Nanomaterials (Basel). 2023 Feb 23;13(5):818. doi: 10.3390/nano13050818. Nanomaterials (Basel). 2023. PMID: 36903697 Free PMC article.
References
-
- Arafune R. Arafune R. Lin C. L. Kawahara K. Tsukahara N. Surf. Sci. 2013;608:297–300. doi: 10.1016/j.susc.2012.10.022. - DOI
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

