Phosphonopeptides containing free phosphonic groups: recent advances
- PMID: 35518575
- PMCID: PMC9055344
- DOI: 10.1039/d0ra04655h
Phosphonopeptides containing free phosphonic groups: recent advances
Abstract
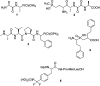
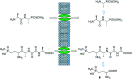
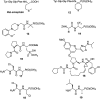
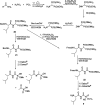
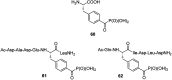
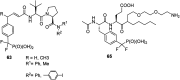
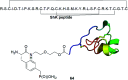
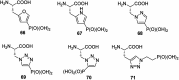
Phosphonopeptides are mimetics of peptides in which phosphonic acid or related (phosphinic, phosphonous etc.) group replaces either carboxylic acid group present at C-terminus, is located in the peptidyl side chain, or phosphonamidate or phosphinic acid mimics peptide bond. Acting as inhibitors of key enzymes related to variable pathological states they display interesting and useful physiologic activities with potential applications in medicine and agriculture. Since the synthesis and biological properties of peptides containing C-terminal diaryl phosphonates and those with phosphonic fragment replacing peptide bond were comprehensively reviewed, this review concentrate on peptides holding free, unsubstituted phosphonic acid moiety. There are two groups of such mimetics: (i) peptides in which aminophosphonic acid is located at C-terminus of the peptide chain with most of them (including antibiotics isolated from bacteria and fungi) exhibiting antimicrobial activity; (ii) non-hydrolysable analogues of phosphonoamino acids, which are useful tools to study physiologic effects of phosphorylations.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures






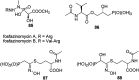

Similar articles
-
Synthetic Methods of Phosphonopeptides.Molecules. 2020 Dec 12;25(24):5894. doi: 10.3390/molecules25245894. Molecules. 2020. PMID: 33322827 Free PMC article. Review.
-
Phosphonic acid: preparation and applications.Beilstein J Org Chem. 2017 Oct 20;13:2186-2213. doi: 10.3762/bjoc.13.219. eCollection 2017. Beilstein J Org Chem. 2017. PMID: 29114326 Free PMC article. Review.
-
Peptide and Pseudopeptide Bond Synthesis in Phosphorus Dipeptide Analogs.Methods Mol Biol. 2020;2103:287-301. doi: 10.1007/978-1-0716-0227-0_20. Methods Mol Biol. 2020. PMID: 31879934
-
Recent synthesis of aminophosphonic acids as potential biological importance.Amino Acids. 2010 Jan;38(1):23-30. doi: 10.1007/s00726-009-0254-7. Epub 2009 Feb 20. Amino Acids. 2010. PMID: 19229586 Review.
-
Facile synthesis of phosphonamidate- and phosphonate-linked phosphonopeptides.J Pept Sci. 2006 Apr;12(4):303-9. doi: 10.1002/psc.727. J Pept Sci. 2006. PMID: 16245363
Cited by
-
Genetic Encoding of Phosphorylated Amino Acids into Proteins.Chem Rev. 2024 May 22;124(10):6592-6642. doi: 10.1021/acs.chemrev.4c00110. Epub 2024 May 1. Chem Rev. 2024. PMID: 38691379 Free PMC article. Review.
-
Phosphorus-Derived Isatin Hydrazones: Synthesis, Structure, Thromboelastography, Antiplatelet, and Anticoagulation Activity Evaluation.Int J Mol Sci. 2025 Jun 26;26(13):6147. doi: 10.3390/ijms26136147. Int J Mol Sci. 2025. PMID: 40649924 Free PMC article.
-
Synthetic Methods of Phosphonopeptides.Molecules. 2020 Dec 12;25(24):5894. doi: 10.3390/molecules25245894. Molecules. 2020. PMID: 33322827 Free PMC article. Review.
-
Enzymatic Synthesis of Biologically Active H-Phosphinic Analogue of α-Ketoglutarate.Biomolecules. 2024 Dec 10;14(12):1574. doi: 10.3390/biom14121574. Biomolecules. 2024. PMID: 39766281 Free PMC article.
-
Dipeptide analogues of fluorinated aminophosphonic acid sodium salts as moderate competitive inhibitors of cathepsin C.Beilstein J Org Chem. 2023 Apr 12;19:434-439. doi: 10.3762/bjoc.19.33. eCollection 2023. Beilstein J Org Chem. 2023. PMID: 37091732 Free PMC article.
References
-
- Aminophosphonic and Aminophosphinic Acids: Chemistry and Biological Activity, ed. V. Kukhar and H. Hudson, John Wiley & Sons, New York, 2000
-
- Ogawa Y. Tsuruoka T. Inouye S. Niida T. Sci. Rep. Meiji Seika Kaisha. 1973;13:42–48.
Publication types
LinkOut - more resources
Full Text Sources

