1-Alkyl-3-alkylindolin-2-imine hydrochlorides as useful building blocks in the copper-catalyzed synthesis of polycyclic indoline scaffolds
- PMID: 35518700
- PMCID: PMC9062002
- DOI: 10.1039/c9ra00995g
1-Alkyl-3-alkylindolin-2-imine hydrochlorides as useful building blocks in the copper-catalyzed synthesis of polycyclic indoline scaffolds
Abstract
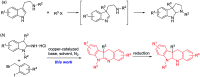
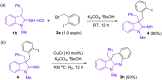
A novel and efficient copper-catalyzed synthesis of dihydro-6H-indolo[2,3-b]quinoline derivatives has been developed by using 3-alkyl-1-alkylindolin-2-imine hydrochlorides as the building blocks. Furthermore, easy reduction of dihydro-6H-indolo[2,3-b]quinolines with diisobutylaluminum hydride provided tetrahydro-6H-indolo[2,3-b]quinoline derivatives in excellent yields. The present method shows some advantages including use of cheap cuprous chloride as the catalyst and tolerance of wide functional groups.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures



Similar articles
-
Highly Enantioselective Iridium-Catalyzed Cascade Double Allylation Strategy: Synthesis of Pyrrolidinoindolines with an All-Carbon Quaternary Stereocenter.Org Lett. 2019 Oct 18;21(20):8501-8505. doi: 10.1021/acs.orglett.9b03382. Epub 2019 Oct 8. Org Lett. 2019. PMID: 31591895
-
An expeditious I2-catalyzed entry into 6H-indolo[2,3-b]quinoline system of cryptotackieine.J Org Chem. 2009 Nov 6;74(21):8369-72. doi: 10.1021/jo901361x. J Org Chem. 2009. PMID: 19788169
-
Efficient two-step synthesis of structurally diverse indolo[2,3-b]quinoline derivatives.Org Biomol Chem. 2019 Feb 27;17(9):2321-2325. doi: 10.1039/c8ob03033b. Org Biomol Chem. 2019. PMID: 30724948
-
6H-Indolo[2,3-b]quinoxalines: DNA and protein interacting scaffold for pharmacological activities.Mini Rev Med Chem. 2013 Aug;13(10):1415-20. doi: 10.2174/13895575113139990005. Mini Rev Med Chem. 2013. PMID: 23701655 Review.
-
Construction of indole- and isoquinoline-fused nitrogen-containing heterocycles through copper-catalyzed multi-component reaction.Yakugaku Zasshi. 2010 Jul;130(7):925-36. doi: 10.1248/yakushi.130.925. Yakugaku Zasshi. 2010. PMID: 20606372 Review.
References
-
- Anthoni U., Christophersen C. and Nielsen P. H., in Alkaloids: Chemical and Biological Perspectives, ed. S. W.Pelletier, Wiley, New York, 1999, vol. 13, p. 163
- Pearce H. L., in The Alkaloids, ed. A.Brossi and M.Suffness, Academic, San Diego, CA, 1990, vol. 37, p. 145
-
- Dalsgaard P. W. Blunt J. W. Munro M. H. G. Frisvad J. C. Christophersen C. J. Nat. Prod. 2005;68:258. doi: 10.1021/np049646l. - DOI - PubMed
- Andersen B. Smedsgaard J. Frisvad J. C. J. Agric. Food Chem. 2004;52:2421. doi: 10.1021/jf035406k. - DOI - PubMed
- Hayashi H. Matsumoto H. Akiyama K. Biosci., Biotechnol., Biochem. 2004;68:753. doi: 10.1271/bbb.68.753. - DOI - PubMed
- Jadulco R. Edrada R. A. Ebel R. Berg A. Schaumann K. Wray V. Steube K. Proksch P. J. Nat. Prod. 2004;67:78. doi: 10.1021/np030271y. - DOI - PubMed
- Numata A. Takahashi C. Ito Y. Takada T. Kawai K. Usami Y. Matsumura E. Imachi M. Ito T. Hasegawa T. Tetrahedron Lett. 1993;34:2355. doi: 10.1016/S0040-4039(00)77612-X. - DOI
-
-
For a recent review, see:
- Trost B. M. Osipov M. Chem. –Eur. J. 2015;21:16318. doi: 10.1002/chem.201501735. - DOI - PMC - PubMed
-
; For selected papers, see:
- Yang J. Wu H. Shen Y. Qin Y. J. Am. Chem. Soc. 2007;129:13794. doi: 10.1021/ja075705g. - DOI - PubMed
- Liu P. Seo J. H. Weinreb S. M. Angew. Chem., Int. Ed. 2010;49:2000. doi: 10.1002/anie.200906818. - DOI - PMC - PubMed
- Seo J. H. Artman III G. D. Weinreb S. M. J. Org. Chem. 2006;71:8891. doi: 10.1021/jo061660a. - DOI - PMC - PubMed
- Belmar J. Funk R. L. J. Am. Chem. Soc. 2012;134:16941. doi: 10.1021/ja307277w. - DOI - PubMed
- Fuchs J. R. Funk R. L. J. Am. Chem. Soc. 2004;126:5068. doi: 10.1021/ja049569g. - DOI - PubMed
- Han S.-J. Vogt F. Krishnan S. May J. A. Gatti M. Virgil S. C. Stoltz B. M. Org. Lett. 2014;16:3316. doi: 10.1021/ol5013263. - DOI - PMC - PubMed
- Han S.-J. Vogt F. May J. A. Krishnan S. Gatti M. Virgil S. C. Stoltz B. M. J. Org. Chem. 2015;80:528. doi: 10.1021/jo502534g. - DOI - PMC - PubMed
- Zuo Z. Xie W. Ma D. J. Am. Chem. Soc. 2010;132:13226. doi: 10.1021/ja106739g. - DOI - PubMed
- Zuo Z. Ma D. Angew. Chem., Int. Ed. 2011;50:12008. doi: 10.1002/anie.201106205. - DOI - PubMed
- Sabahi A. Novikov A. Rainier J. D. Angew. Chem., Int. Ed. 2006;45:4317. doi: 10.1002/anie.200601278. - DOI - PubMed
-
LinkOut - more resources
Full Text Sources

