Retro Diels Alder protocol for regioselective synthesis of novel [1,2,4]triazolo[4,3- a]pyrimidin-7(1 H)-ones
- PMID: 35519032
- PMCID: PMC9056728
- DOI: 10.1039/d0ra04345a
Retro Diels Alder protocol for regioselective synthesis of novel [1,2,4]triazolo[4,3- a]pyrimidin-7(1 H)-ones
Abstract
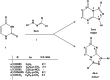
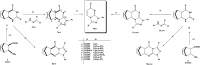
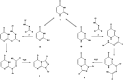
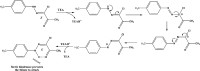
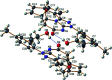
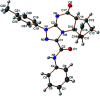
Reactions of diastereochemically varied norbornene-condensed 2-thioxopyrimidin-4-ones 6 and 10 with variously functionalized hydrazonoyl chlorides 2a-h gave regioselectively angular norbornene-based [1,2,4]triazolo[4,3-a]pyrimidin-7(1H)-ones 7a-h and 11a,c-e, respectively. Thermal retro Diels-Alder (RDA) reaction of 7a-h and 11a,c-e resulted in the target compounds 4a-h as single products. On the other hand, reactions of thiouracil 1 and hydrozonoyl chlorides 2a-e gave regioselectively [1,2,4]triazolo[4,3-a]pyrimidinone-5(1H)-ones 3a-e. The opposite regioselectivity of thiouracil 1 and norbornene-condensed 2-thioxopyrimidin-4-ones 6 and 10 was attributed to electronic factors according to DFT calculations. The angular structure of norbornene based [1,2,4]triazolo[4,3-a]pyrimidin-7(1H)-ones was confirmed by single crystal X-ray crystallography.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures

Similar articles
-
Angular Regioselectivity in the Reactions of 2-Thioxopyrimidin-4-ones and Hydrazonoyl Chlorides: Synthesis of Novel Stereoisomeric Octahydro[1,2,4]triazolo[4,3-a]quinazolin-5-ones.Molecules. 2020 Dec 1;25(23):5673. doi: 10.3390/molecules25235673. Molecules. 2020. PMID: 33271898 Free PMC article.
-
Angular Regioselective Synthesis of Varied Functionalized Hexahydro-1,2,4-triazolo[4,3-a]quinazolin-9-ones and Their Antiproliferative Action.Molecules. 2023 Apr 25;28(9):3718. doi: 10.3390/molecules28093718. Molecules. 2023. PMID: 37175130 Free PMC article.
-
A Facile One-pot Synthesis and Antimicrobial Activity of Pyrido [2,3-d][1,2,4]triazolo[4,3-a]pyrimidin-5-ones.Acta Chim Slov. 2011 Mar;58(1):87-94. Acta Chim Slov. 2011. PMID: 24061948
-
Domino retro Diels-Alder/Diels-Alder reaction: an efficient protocol for the synthesis of highly functionalized bicyclo[2.2.2]octenones and bicyclo[2.2.2]octadienones.Org Biomol Chem. 2006 Jun 7;4(11):2267-77. doi: 10.1039/b602928k. Epub 2006 May 4. Org Biomol Chem. 2006. PMID: 16729136
-
Application of the aza-Diels-Alder reaction in the synthesis of natural products.Org Biomol Chem. 2017 Apr 11;15(15):3105-3129. doi: 10.1039/c6ob02761j. Org Biomol Chem. 2017. PMID: 28327756 Review.
Cited by
-
Angular Regioselectivity in the Reactions of 2-Thioxopyrimidin-4-ones and Hydrazonoyl Chlorides: Synthesis of Novel Stereoisomeric Octahydro[1,2,4]triazolo[4,3-a]quinazolin-5-ones.Molecules. 2020 Dec 1;25(23):5673. doi: 10.3390/molecules25235673. Molecules. 2020. PMID: 33271898 Free PMC article.
-
Angular Regioselective Synthesis of Varied Functionalized Hexahydro-1,2,4-triazolo[4,3-a]quinazolin-9-ones and Their Antiproliferative Action.Molecules. 2023 Apr 25;28(9):3718. doi: 10.3390/molecules28093718. Molecules. 2023. PMID: 37175130 Free PMC article.
References
-
- Dong H. Gao Z. Li R. Hu Y. Dong H. Xie Z. RSC Adv. 2014;4:55827. doi: 10.1039/C4RA02714K. - DOI
-
- Jafari B. Yelibayeva N. Ospanov M. Ejaz S. A. Afzal S. Khan S. U. Abilov Z. A. Turmukhanova M. Z. Kalugin S. N. Safarov S. Lecka J. Sévigny J. Rahman Q. Ehlers P. Iqbal J. Langer P. RSC Adv. 2016;6:107556. doi: 10.1039/C6RA22750C. - DOI
-
- Dinakaran V. S. Bomma B. Srinivasan K. K. Der Pharma Chem. 2012;4:255.
-
- Mishra R. Tomar I. Int. J. Pharm. Sci. Res. 2011;2:758.
-
- Fares M. Abou-Seri S. M. Abdel-Aziz H. A. Abbas S. E. S. Youssef M. M. Eladwy R. A. Eur. J. Med. Chem. 2014;83:155. doi: 10.1016/j.ejmech.2014.06.027. - DOI - PubMed
- Abdelhamid A. O. Gomha S. M. Abdelriheem N. A. Kandeel S. M. Molecules. 2016;21:929. doi: 10.1016/j.ejmech.2014.06.027. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

