Recent advances and future perspectives of sol-gel derived porous bioactive glasses: a review
- PMID: 35519068
- PMCID: PMC9056785
- DOI: 10.1039/d0ra04287k
Recent advances and future perspectives of sol-gel derived porous bioactive glasses: a review
Abstract
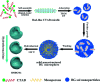
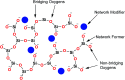
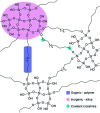
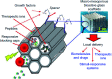
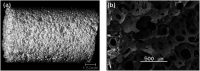
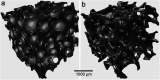
The sol-gel derived porous bioactive glasses have drawn worldwide attention by virtue of the convenience and flexibility of this versatile synthesis method. In this review, the recent advances in sol-gel processed porous bioactive glasses in biomedical fields, especially for bone tissue regeneration applications have been comprehensively reviewed. Generally, it is envisaged that the morphology and chemical compositions of sol-gel derived porous bioactive glasses significantly affect their biological properties. Therefore, the controlled synthesis of these porous glasses is critical to their effective use in the biomedical fields. With this context, the first part of the review briefly describes the fundamentals of the sol-gel technique. In the subsequent section, different approaches frequently used for the sol-gel synthesis of porous glasses such as microemulsion and acid-catalyzed based synthesis have been reviewed. In the later part of the review, different types of sol-gel derived bioactive glasses namely silica, phosphate and silica-titania based glasses along with organic-inorganic hybrids materials have been discussed. The review also discusses the chemical, surface, mechanical and biological properties and further highlights the strategies to control the pore structure, shape, size and compositions of sol-gel derived bioactive glasses. Finally, the review provides a detailed discussion about the bone tissue regeneration application of different types of sol-gel derived bioactive glasses and presents future research perspectives.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





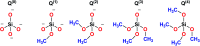













Similar articles
-
Effect of calcium source on structure and properties of sol-gel derived bioactive glasses.Langmuir. 2012 Dec 18;28(50):17465-76. doi: 10.1021/la303768b. Epub 2012 Dec 6. Langmuir. 2012. PMID: 23171477
-
Composite Biomaterials Based on Sol-Gel Mesoporous Silicate Glasses: A Review.Bioengineering (Basel). 2017 Feb 23;4(1):15. doi: 10.3390/bioengineering4010015. Bioengineering (Basel). 2017. PMID: 28952496 Free PMC article. Review.
-
Fabrication, structure and biological properties of organic acid-derived sol-gel bioactive glasses.Biomed Mater. 2010 Oct;5(5):054103. doi: 10.1088/1748-6041/5/5/054103. Epub 2010 Sep 28. Biomed Mater. 2010. PMID: 20876955
-
Sol-gel silica-based biomaterials and bone tissue regeneration.Acta Biomater. 2010 Aug;6(8):2874-88. doi: 10.1016/j.actbio.2010.02.012. Epub 2010 Feb 10. Acta Biomater. 2010. PMID: 20152946 Review.
-
Indirect rapid prototyping of sol-gel hybrid glass scaffolds for bone regeneration - Effects of organic crosslinker valence, content and molecular weight on mechanical properties.Acta Biomater. 2016 Apr 15;35:318-29. doi: 10.1016/j.actbio.2016.02.038. Epub 2016 Feb 27. Acta Biomater. 2016. PMID: 26925964
Cited by
-
Sol-Gel ceramic glazes with photocatalytic activity.J Solgel Sci Technol. 2022;102(3):535-549. doi: 10.1007/s10971-022-05787-z. Epub 2022 Apr 23. J Solgel Sci Technol. 2022. PMID: 35494725 Free PMC article.
-
Properties and antibacterial effectiveness of metal-ion doped borate-based bioactive glasses.Future Microbiol. 2025 Mar;20(4):315-331. doi: 10.1080/17460913.2025.2470029. Epub 2025 Mar 13. Future Microbiol. 2025. PMID: 40079871 Review.
-
Multifunctional Sr,Mg-Doped Mesoporous Bioactive Glass Nanoparticles for Simultaneous Bone Regeneration and Drug Delivery.Int J Mol Sci. 2024 Jul 24;25(15):8066. doi: 10.3390/ijms25158066. Int J Mol Sci. 2024. PMID: 39125634 Free PMC article.
-
Diffusion/Reaction Limited Aggregation Approach for Microstructure Evolution and Condensation Kinetics during Synthesis of Silica-Based Alcogels.Int J Mol Sci. 2023 Jan 19;24(3):1999. doi: 10.3390/ijms24031999. Int J Mol Sci. 2023. PMID: 36768318 Free PMC article.
-
Macroporosity Control by Phase Separation in Sol-Gel Derived Monoliths and Microspheres.Materials (Basel). 2021 Jul 29;14(15):4247. doi: 10.3390/ma14154247. Materials (Basel). 2021. PMID: 34361442 Free PMC article. Review.
References
-
- Owens G. J. Singh R. K. Foroutan F. Alqaysi M. Han C. M. Mahapatra C. Kim H. W. Sol–gel based materials for biomedical applications. Prog. Mater. Sci. 2016;77:1–79. doi: 10.1016/j.pmatsci.2015.12.001. - DOI
-
- Tan S. N. Wang W. Ge L. 3.30 Biosensors based on sol–gel derived materials. Compr. Biomater. II. 2017;3:657–689.
-
- Monton M. R. N. Forsberg E. M. Brennan J. D. Tailoring sol–gel derived silica materials for optical biosensing. Chem. Mater. 2012;24:796–811. doi: 10.1021/cm202798e. - DOI
-
- Akpan U. G. Hameed B. H. The advancement in sol–gel method of doped TiO2 photocatalyst. Appl. Catal., A. 2010;375:1–11. doi: 10.1016/j.apcata.2009.12.023. - DOI
-
- Levy D. and Zayat M., The sol–gel handbook, ed. D. Levy and M. Zayat, vol. 1–3, Wiley VCH, USA, 2015, pp. 1–1508
Publication types
LinkOut - more resources
Full Text Sources

