Interplay of π-stacking and inter-stacking interactions in two-component crystals of neutral closed-shell aromatic compounds: periodic DFT study
- PMID: 35519116
- PMCID: PMC9055576
- DOI: 10.1039/d0ra04799f
Interplay of π-stacking and inter-stacking interactions in two-component crystals of neutral closed-shell aromatic compounds: periodic DFT study
Abstract
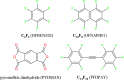
This paper bridges the gap between high-level ab initio computations of gas-phase models of 1 : 1 arene-arene complexes and calculations of the two-component (binary) organic crystals using atom-atom potentials. The studied crystals consist of electron-rich and electron-deficient compounds, which form infinite stacks (columns) of heterodimers. The sublimation enthalpy of crystals has been evaluated by DFT periodic calculations, while intermolecular interactions have been characterized by Bader analysis of the periodic electronic density. The consideration of aromatic compounds without a dipole moment makes it possible to reveal the contribution of quadrupole-quadrupole interactions to the π-stacking energy. These interactions are significant for heterodimers formed by arenes with more than 2 rings, with absolute values of the traceless quadrupole moment (Q zz) larger than 10 D Å. The further aggregation of neighboring stacks is due to the C-H⋯F interactions in arene/perfluoroarene crystals. In crystals consisting of arene and an electron-deficient compound such as pyromellitic dianhydride, aggregation occurs due to the C-H⋯O interactions. The C-H⋯F and C-H⋯O inter-stacking interactions make the main contribution to the sublimation enthalpy, which exceeds 150 kJ mol-1 for the two-component crystals formed by arenes with more than 2 rings.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures



References
-
- Sutton C. Risko C. Brédas J.-L. Noncovalent Intermolecular Interactions in Organic Electronic Materials: Implications for the Molecular Packing vs. Electronic Properties of Acenes. Chem. Mater. 2016;28:3–16. doi: 10.1021/acs.chemmater.5b03266. - DOI
-
- Mandal A. Choudhury A. Iyer P. K. Mal P. Charge Transfer Versus Arene–Perfluoroarene Interactions in Modulation of Optical and Conductivity Properties in Cocrystals of 2,7-Di-tert-butylpyrene. J. Phys. Chem. C. 2019;123:18198–18206. doi: 10.1021/acs.jpcc.9b03827. - DOI
-
- Chen T. Li M. Liu J. π–π Stacking Interaction: A Nondestructive and Facile Means in Material Engineering for Bioapplications. Cryst. Growth Des. 2018;18:2765–2783.
-
- Jia Z. Xu G. Li Q. Zhang Y. Liu B. Pan Y. Wang X. Wang T. Li F. Boosting the efficiency and stability of polymer solar cells using poly(3-hexylthiophene)-based all-conjugated diblock copolymers containing pentafluorophenyl groups. J. Mater. Sci.: Mater. Electron. 2018;29:10337–10345.
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

