Highly dispersed Ni nanoparticles on mesoporous silica nanospheres by melt infiltration for transfer hydrogenation of aryl ketones
- PMID: 35519300
- PMCID: PMC9064007
- DOI: 10.1039/c9ra01608b
Highly dispersed Ni nanoparticles on mesoporous silica nanospheres by melt infiltration for transfer hydrogenation of aryl ketones
Abstract
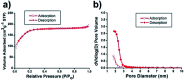
Nickel-based catalysts have been applied to the catalytic reactions for transfer hydrogenation of carbonyl compounds. In the present work, highly dispersed nickel particles located at the pores of mesoporous silica spheres (Ni@mSiO2) were prepared via an optimized melt infiltration route. The nickel nanoparticles of 10 wt% in the Ni@mSiO2 catalyst could be uniformly loaded with high dispersion of 36.3%, resulting excellent performance for catalytic transfer hydrogenation of aryl ketones.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures



Similar articles
-
Ru-Ni Alloy Nanoparticles Loaded on N-Doped Amphiphilic Mesoporous Hollow Carbon@silica Spheres as Catalyst for the Hydrogenation of α-Pinene to cis-Pinane.Chempluschem. 2023 Mar;88(3):e202200443. doi: 10.1002/cplu.202200443. Chempluschem. 2023. PMID: 36807893
-
Formation Combined with Intercalation of Ni and Its Alloy Nanoparticles within Mesoporous Silica for Robust Catalytic Reactions.ACS Appl Mater Interfaces. 2018 Sep 5;10(35):29435-29447. doi: 10.1021/acsami.8b07896. Epub 2018 Aug 22. ACS Appl Mater Interfaces. 2018. PMID: 30089361
-
Revealing the anti-sintering phenomenon on silica-supported nickel catalysts during CO2 hydrogenation.J Environ Sci (China). 2024 Jun;140:270-278. doi: 10.1016/j.jes.2023.08.028. Epub 2023 Sep 2. J Environ Sci (China). 2024. PMID: 38331507
-
Ni Nanoparticles Supported on Cage-Type Mesoporous Silica for CO2 Hydrogenation with High CH4 Selectivity.ChemSusChem. 2016 Sep 8;9(17):2326-31. doi: 10.1002/cssc.201600710. Epub 2016 Aug 17. ChemSusChem. 2016. PMID: 27531065
-
Recent advances in osmium-catalyzed hydrogenation and dehydrogenation reactions.Acc Chem Res. 2015 Feb 17;48(2):363-79. doi: 10.1021/ar5003818. Epub 2015 Feb 4. Acc Chem Res. 2015. PMID: 25650714 Review.
References
-
- Evans D. A. Nelson S. G. Gagne M. R. Muci A. R. J. Am. Chem. Soc. 1993;115:9800–9801. doi: 10.1021/ja00074a057. - DOI
-
- Corey E. J. Helal C. J. Tetrahedron Lett. 1996;37:5675–5678. doi: 10.1016/0040-4039(96)01198-7. - DOI
-
- Alonso F., Nickel Nanoparticles in the Transfer Hydrogenation of Functional Groups, in Met. Nanoparticles Catal. Adv. Appl., The Royal Society of Chemistry, 2014, ch. 5, pp. 83–98
LinkOut - more resources
Full Text Sources

