Substituted adenine quartets: interplay between substituent effect, hydrogen bonding, and aromaticity
- PMID: 35520336
- PMCID: PMC9054646
- DOI: 10.1039/d0ra04585c
Substituted adenine quartets: interplay between substituent effect, hydrogen bonding, and aromaticity
Abstract
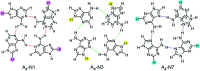
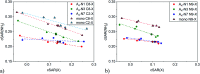
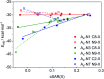
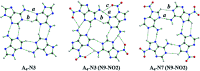
Adenine, one of the components of DNA/RNA helices, has the ability to form self-organizing structures with cyclic hydrogen bonds (A4), similar to guanine quartets. Here, we report a computational investigation of the effect of substituents (X = NO2, Cl, F, H, Me, and NH2) on the electronic structure of 9H-adenine and its quartets (A4-N1, A4-N3, and A4-N7). DFT calculations were used to show the relationships between the electronic nature of the substituents, strength of H-bonds in the quartets, and aromaticity of five- and six-membered rings of adenine. We demonstrated how the remote substituent X modifies the proton-donating properties of the NH2 group involved in the H-bonds within quartets and how the position of the substituent and its electronic nature affect the stability of the quartets. We also showed the possible changes in electronic properties of the substituent and aromaticity of adenine rings caused by tetramer formation. The results indicate that the observed relationships depend on the A4 type. Moreover, the same substituent can both strengthen and weaken intermolecular interactions, depending on the substitution position.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures

Similar articles
-
Mutual Relations between Substituent Effect, Hydrogen Bonding, and Aromaticity in Adenine-Uracil and Adenine-Adenine Base Pairs.Molecules. 2020 Aug 13;25(16):3688. doi: 10.3390/molecules25163688. Molecules. 2020. PMID: 32823565 Free PMC article.
-
Impact of the Substituents on the Electronic Structure of the Four Most Stable Tautomers of Purine and Their Adenine Analogues.ACS Omega. 2020 May 12;5(20):11570-11577. doi: 10.1021/acsomega.0c00820. eCollection 2020 May 26. ACS Omega. 2020. PMID: 32478247 Free PMC article.
-
Proton Changes the Rules of the Game: Insight into the Nature of "Proton Sponges".Chemphyschem. 2025 May 5;26(9):e202400972. doi: 10.1002/cphc.202400972. Epub 2025 Mar 6. Chemphyschem. 2025. PMID: 39929770
-
Effect of the substituent and hydrogen bond on the geometry and electronic properties of OH and O(-) groups in para-substituted phenol and phenolate derivatives.J Phys Chem A. 2010 Oct 14;114(40):10885-90. doi: 10.1021/jp1071204. J Phys Chem A. 2010. PMID: 20853885
-
Synthetic techniques for thermodynamically disfavoured substituted six-membered rings.Nat Rev Chem. 2024 Jul;8(7):535-550. doi: 10.1038/s41570-024-00612-3. Epub 2024 May 31. Nat Rev Chem. 2024. PMID: 38822206 Review.
Cited by
-
Mutual Relations between Substituent Effect, Hydrogen Bonding, and Aromaticity in Adenine-Uracil and Adenine-Adenine Base Pairs.Molecules. 2020 Aug 13;25(16):3688. doi: 10.3390/molecules25163688. Molecules. 2020. PMID: 32823565 Free PMC article.
-
Unveiling Adenine H‑bonded Hexads: Hierarchical Self-Assembly for Helical Columnar Functional Materials.JACS Au. 2025 Jun 19;5(7):3115-3124. doi: 10.1021/jacsau.5c00256. eCollection 2025 Jul 28. JACS Au. 2025. PMID: 40747067 Free PMC article.
-
Analysis of Conformational, Structural, Magnetic, and Electronic Properties Related to Antioxidant Activity: Revisiting Flavan, Anthocyanidin, Flavanone, Flavonol, Isoflavone, Flavone, and Flavan-3-ol.ACS Omega. 2021 Mar 24;6(13):8908-8918. doi: 10.1021/acsomega.0c06156. eCollection 2021 Apr 6. ACS Omega. 2021. PMID: 33842761 Free PMC article.
References
-
- Huang M. C. Montgomery J. A. Thorpe M. C. Stewart E. L. III Secrist J. A. Blakley R. L. Formation of 3-(2′-Deoxyribofuranosyl) and 9-(2′-Deoxyribofuranosyl) Nucleosides of 8-Substituted Purines by Nucleoside Deoxyribosyltransferase. Arch. Biochem. Biophys. 1983;222:133–144. doi: 10.1016/0003-9861(83)90510-6. - DOI - PubMed
-
- Fonseca Guerra C. van der Wijst T. Bickelhaupt F. M. Substituent Effects on Hydrogen Bonding in Watson–Crick Base Pairs. A Theoretical Study. Struct. Chem. 2005;16:211–221. doi: 10.1007/s11224-005-4453-x. - DOI
LinkOut - more resources
Full Text Sources

