Bifunctional nano-Ag3PO4 with capabilities of enhancing ceftazidime for sterilization and removing residues
- PMID: 35520599
- PMCID: PMC9064663
- DOI: 10.1039/c9ra01969c
Bifunctional nano-Ag3PO4 with capabilities of enhancing ceftazidime for sterilization and removing residues
Abstract
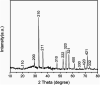
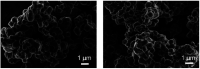
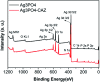
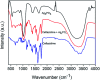
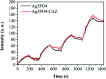
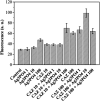
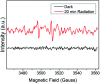
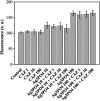
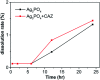
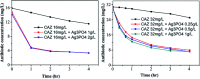
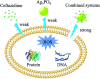
Since the efficacy of antibiotics towards bacteria is decreasing over time, the rising of antibiotic emission has become an increasingly grave issue. In this study, we proposed an integrated antibacterial nanotechnology without pollution residues, which synergistically enhances the antibacterial activity of ceftazidime by using the inorganic nano-Ag3PO4, and subsequently removes drug residues by photocatalysis. Ag3PO4 were synthesized using a simple ion-exchange method without any reducing agent or protectant. The combined antibacterial activity of Ag3PO4 and 22 kinds of antibiotics against Escherichia coli was first studied. The results showed that Ag3PO4 and ceftazidime exhibited the best synergistic effect. Next, the synergy mechanism was proposed, the non-chemical bond forces between Ag3PO4 and ceftazidime was determined by zeta potential analyzer, X-ray photoelectron spectroscopy (XPS) and infrared spectroscopy (IR). The interaction between antimicrobials and bacteria was further demonstrated by surface plasma resonance spectroscopy (SPR), scanning electron microscopy (SEM) and propidium iodide (PI) staining. In addition, the production of reactive oxygen species (ROS), the induction of oxidative stress and dissolution of silver ions in Ag3PO4 were studied and found out that only under light, could the ROS be generated. In conclusion, the synergistic effect of Ag3PO4 and ceftazidime is responsible for the joint destruction of cell wall.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures

References
-
- Singh S. Gundampati R. K. Mitra K. Ramesh K. Jagannadham M. V. Misra N. Ray B. RSC Adv. 2015;5:81994–82004. doi: 10.1039/C5RA13286J. - DOI
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

