Single-cell-resolution map of human retinal pigment epithelium helps discover subpopulations with differential disease sensitivity
- PMID: 35522714
- PMCID: PMC9171647
- DOI: 10.1073/pnas.2117553119
Single-cell-resolution map of human retinal pigment epithelium helps discover subpopulations with differential disease sensitivity
Abstract
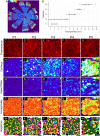
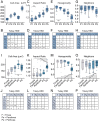
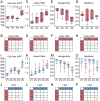
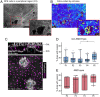
Regional phenotypic and functional differences in the retinal pigment epithelium (RPE) monolayer have been suggested to account for regional susceptibility in ocular diseases such as age-related macular degeneration (AMD), late-onset retinal degeneration (L-ORD), and choroideremia (CHM). However, a comprehensive description of human topographical RPE diversity is not yet available, thus limiting the understanding of regional RPE diversity and degenerative disease sensitivity in the eye. To develop a complete morphometric RPE map of the human eye, artificial intelligence–based software was trained to recognize, segment, and analyze RPE borders. Five statistically different, concentric RPE subpopulations (P1 to P5) were identified using cell area as a parameter, including a subpopulation (P4) with cell area comparable to that of macular cells in the far periphery of the eye. This work provides a complete reference map of human RPE subpopulations and their location in the eye. In addition, the analysis of cadaver non-AMD and AMD eyes and ultra-widefield fundus images of patients revealed differential vulnerability of the five RPE subpopulations to different retinal diseases.
Keywords: AMD; artificial intelligence; cell morphometry; data science; retinal degeneration.
Conflict of interest statement
The authors declare no competing interest.
Figures


References
-
- Kuntz C. A., et al. , Sub-retinal pigment epithelial deposits in a dominant late-onset retinal degeneration. Invest. Ophthalmol. Vis. Sci. 37, 1772–1782 (1996). - PubMed
-
- Jacobson S. G., Cideciyan A. V., Wright E., Wright A. F., Phenotypic marker for early disease detection in dominant late-onset retinal degeneration. Invest. Ophthalmol. Vis. Sci. 42, 1882–1890 (2001). - PubMed
-
- Styles C. J., Dhillon B., Wright A. F., The diagnosis of autosomal dominant late-onset retinal degeneration in two sisters. Eye 17, 530–532 (2003). - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

