Effects of 3 months of multi-nutrient supplementation on the immune system and muscle and respiratory function of older adults in aged care (The Pomerium Study): protocol for a randomised controlled trial
- PMID: 35523505
- PMCID: PMC9082724
- DOI: 10.1136/bmjopen-2021-059075
Effects of 3 months of multi-nutrient supplementation on the immune system and muscle and respiratory function of older adults in aged care (The Pomerium Study): protocol for a randomised controlled trial
Abstract
Introduction: Immunosenescence leads to increased morbidity and mortality associated with viral infections and weaker vaccine responses. This has been well documented for seasonal influenza and the current pandemic with SARS-CoV-2 (COVID-19), which disproportionately impact older adults, particularly those in residential aged care facilities. Inadequate nutrient intakes associated with impaired immunity, respiratory and muscle function are likely to augment the effects of immunosenescence. In this study, we test whether the impact of inadequate nutrition can be reversed using multi-nutrient supplementation, consequently enhancing vaccine responses, reducing the risk of viral infections and improving respiratory and muscle function.
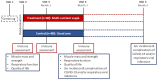
Methods and analysis: The Pomerium Study is a 3-month, single-blind, randomised, controlled trial testing the effects of two daily servings of an oral multi-nutrient supplement (330 kcal, 20 g protein, 1.5 g calcium 3-hydroxy-3-methylbutyrate monohydrate (CaHMB), 449 mg calcium, 500 IU vitamin D3 and 25 vitamins and minerals) on the immune system and muscle and respiratory function of older adults in aged care in Melbourne, Australia. 160 older adults (≥75 years old) will be recruited from aged care facilities and randomised to treatment (multi-nutrient supplement) or control (usual care). The primary outcome is a change in T-cell subsets CD8 + and CD28null counts at months 1 and 3. Secondary outcomes measured at baseline and month 3 are multiple markers of immunosenescence (also at 1 month), body composition (bioimpedance), handgrip strength (dynamometer), physical function (short physical performance battery), respiratory function (spirometry) and quality of life (EQ-5D-5L). Incidence and complications of COVID-19 and/or viral infections (ie, hospitalisation, complications or death) will be recorded throughout the trial, including 3 months after supplementation is ceased.
Ethics and dissemination: This study was approved by Melbourne Health Human Research Ethics Committee (Ref No. HREC/73985/MH-2021, ERM Ref No. RMH73985, Melbourne Health Site Ref No. 2021.115). Written informed consent will be obtained from participants. Results will be published in peer-reviewed journals and made available to key aged care stakeholders, including providers, residents, and government bodies.
Trial registration number: ACTRN12621000420842.
Keywords: COVID-19; GERIATRIC MEDICINE; IMMUNOLOGY; INFECTIOUS DISEASES; NUTRITION & DIETETICS.
© Author(s) (or their employer(s)) 2022. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: GD and SI-B serve as members of the Advisory Board of Abbott Australia. GD serves on the Scientific Advisory Board at TSI Pharmaceuticals. BK is currently supported by a research fellowship from TSI Pharmaceuticals. All the other authors declare that they have no competing interests.
Similar articles
-
Safety and Efficacy of Imatinib for Hospitalized Adults with COVID-19: A structured summary of a study protocol for a randomised controlled trial.Trials. 2020 Oct 28;21(1):897. doi: 10.1186/s13063-020-04819-9. Trials. 2020. PMID: 33115543 Free PMC article.
-
Effect of targeted nutrient supplementation on physical activity and health-related quality of life in COPD: study protocol for the randomised controlled NUTRECOVER trial.BMJ Open. 2022 Mar 16;12(3):e059252. doi: 10.1136/bmjopen-2021-059252. BMJ Open. 2022. PMID: 35296491 Free PMC article.
-
Controlled, double-blind, randomized trial to assess the efficacy and safety of hydroxychloroquine chemoprophylaxis in SARS CoV2 infection in healthcare personnel in the hospital setting: A structured summary of a study protocol for a randomised controlled trial.Trials. 2020 Jun 3;21(1):472. doi: 10.1186/s13063-020-04400-4. Trials. 2020. PMID: 32493494 Free PMC article.
-
Do Diet and Dietary Supplements Mitigate Clinical Outcomes in COVID-19?Nutrients. 2022 May 2;14(9):1909. doi: 10.3390/nu14091909. Nutrients. 2022. PMID: 35565876 Free PMC article. Review.
-
The aging of the immune system and its implications for transplantation.Geroscience. 2023 Jun;45(3):1383-1400. doi: 10.1007/s11357-022-00720-2. Epub 2023 Jan 10. Geroscience. 2023. PMID: 36626019 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous