Simultaneous Photodynamic Eradication of Tooth Biofilm and Tooth Whitening with an Aggregation-Induced Emission Luminogen
- PMID: 35524635
- PMCID: PMC9284169
- DOI: 10.1002/advs.202106071
Simultaneous Photodynamic Eradication of Tooth Biofilm and Tooth Whitening with an Aggregation-Induced Emission Luminogen
Abstract
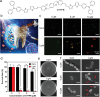
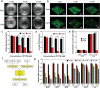
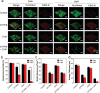
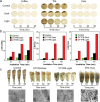
Dental caries is among the most prevalent dental diseases globally, which arises from the formation of microbial biofilm on teeth. Besides, tooth whitening represents one of the fastest-growing areas of cosmetic dentistry. It will thus be great if tooth biofilm eradication can be combined with tooth whitening. Herein, a highly efficient photodynamic dental therapy strategy is reported for tooth biofilm eradication and tooth discoloration by employing a photosensitizer (DTTPB) with aggregation-induced emission characteristics. DTTPB can efficiently inactivate S. mutans, and inhibit biofilm formation by suppressing the expression of genes associated with extracellular polymeric substance synthesis, bacterial adhesion, and superoxide reduction. Its inhibition performance can be further enhanced through combined treatment with chlorhexidine. Besides, DTTPB exhibits an excellent tooth-discoloration effect on both colored saliva-coated hydroxyapatite and clinical teeth, with short treatment time (less than 1 h), better tooth-whitening performance than 30% hydrogen peroxide, and almost no damage to the teeth. DTTPB also demonstrates excellent biocompatibility with neglectable hemolysis effect on mouse red blood cells and almost no killing effect on mammalian cells, which enables its potential applications for simultaneous tooth biofilm eradication and tooth whitening in clinical dentistry.
Keywords: Streptococcus mutans; aggregation-induced emission; biofilm; photodynamic therapy; tooth whitening.
© 2022 The Authors. Advanced Science published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Peres M. A., Macpherson L. M. D., Weyant R. J., Daly B., Venturelli R., Mathur M. R., Listl S., Celeste R. K., Guarnizo‐Herreño C. C., Kearns C., Benzian H., Allison P., Watt R. G., Lancet 2019, 394, 249. - PubMed
-
- Vos T., Allen C., Arora M., Barber R. M., Brown A., Carter A., Casey D. C., Charlson F. J., Chen A. Z., Coggeshall M., Cornaby L., Dandona L., Dicker D. J., Dilegge T., Erskine H. E., Ferrari A. J., Fitzmaurice C., Fleming T., Forouzanfar M. H., Fullman N., Goldberg E. M., Graetz N., Haagsma J. A., Hay S. I., Johnson C. O., Kassebaum N. J., Kawashima T., Kemmer L., Khalil I. A., Kyu H. H., et al., Lancet 2016, 388, 1545.
-
- Helöe L. A., Haugejorden O., Community Dent. Oral Epidemiol. 1981, 9, 294. - PubMed
-
- Marsh P. D., Microbiology 2003, 149, 279. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
