The Role of Mitochondria-Targeting miRNAs in Intracerebral Hemorrhage
- PMID: 35524670
- PMCID: PMC10286585
- DOI: 10.2174/1570159X20666220507021445
The Role of Mitochondria-Targeting miRNAs in Intracerebral Hemorrhage
Abstract
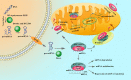
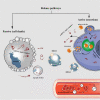
Non-traumatic intracerebral hemorrhage (ICH) is the most common type of hemorrhagic stroke, most often occurring between the ages of 45 and 60. Arterial hypertension (AH) is most often the cause of ICH, followed by atherosclerosis, blood diseases, inflammatory changes in cerebral vessels, intoxication and vitamin deficiencies. Cerebral hemorrhage can occur by diapedesis or as a result of a ruptured vessel. AH is difficult to treat, requires surgery and can lead to disability or death. One of the important directions in the study of the pathogenesis of ICH is mitochondrial dysfunction and its regulation. The key role of mitochondrial dysfunction in AH and atherosclerosis, as well as in the development of brain damage after hemorrhage, has been acknowledged. MicroRNAs (miRNAs) are a class of non-coding RNAs (about 18-22 nucleotides) that regulate a variety of biological processes including cell differentiation, proliferation, apoptosis, etc., primarily through gene repression. There is growing evidence to support dysregulated miRNAs in various cardiovascular diseases, including ICH. Further, the realization of miRNAs within mitochondrial compartment has challenged the traditional knowledge of signaling pathways involved in the regulatory network of cardiovascular diseases. However, the role of miRNAs in mitochondrial dysfunction for ICH is still under-appreciated, with comparatively much lesser studies and investigations reported, than those in other cardiovascular diseases. In this review, we summarize the up-to-date findings on the published role miRNAs in mitochondrial function for ICH, and the potential use of miRNAs in clinical settings, such as potential therapeutic targets and non-invasive diagnostic/prognostic biomarker tools.
Keywords: Intracerebral hemorrhage; biomarker; miRNA; mitochondria; mitochondrial dysfunction; pathogenesis; therapeutic target.
Copyright© Bentham Science Publishers; For any queries, please email at epub@benthamscience.net.
Conflict of interest statement
The authors declare no conflict of interest, financial or otherwise.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

