Anterior chamber enhancement predicts optic nerve infiltration in retinoblastoma
- PMID: 35524782
- PMCID: PMC9668776
- DOI: 10.1007/s00330-022-08778-4
Anterior chamber enhancement predicts optic nerve infiltration in retinoblastoma
Abstract
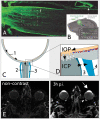
Objectives: As described recently, intravenously injected gadolinium-based contrast agent (GBCA) penetrates into the anterior eye chamber (AC) and is drained from the retina to the distal optic nerve (ON) along perivascular spaces, which serves retinal homeostasis and was termed the orbital glymphatic system (GS). Independently, AC enhancement predicted ON infiltration, a major risk factor for advanced retinoblastoma (RB), in a small RB patient cohort. We aimed to review the supposed imaging biomarker for ON infiltration in a large RB cohort and with respect to the recently described orbital GS.
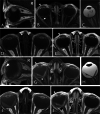
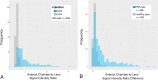
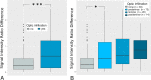
Methods: This IRB-approved retrospective single-center study encompassed 539 orbital MRIs performed with an orbital coil and with the children under general anesthesia. Differences of signal intensity ratios (∆SIRs) of the AC to the lens were determined between non-contrast and GBCA-enhanced T1-weighted images and were correlated with histopathologic presence of ON infiltration.
Results: ∆SIR of the RB eye was an independent, significant predictor for ON invasion in multivariate analysis with adjustment for tumor size (p < 0.05) and increased with infiltration level.
Conclusions: GBCA enhancement of the AC predicts ON infiltration. This might be caused by impairment of the orbital glymphatic system, which is supposed to clear toxic metabolites from the retina to the postlaminar ON. In RB with ON infiltration, this efflux path is likely to be inhibited, which is supposed to result in disturbed retinal homeostasis, release of vascular endothelial growth factor, and iris neovascularization, which increases penetration of GBCA into the AC.
Key points: • Infiltration of the optic nerve can be predicted by anterior chamber enhancement after intravenous MRI contrast agent administration. • Increased anterior chamber enhancement in retinoblastoma with optic nerve infiltration might result from dysfunction of the orbital glymphatic system with disturbance of retinal homeostasis and consecutive iris neovascularization.
Keywords: Gadolinium; Glymphatic system; Magnetic resonance imaging; Optic nerve; Retinoblastoma.
© 2022. The Author(s).
Conflict of interest statement
The authors of this manuscript declare relationships with the following companies: K. Deike-Hofmann: lecture payment by GE, financial study support: Bayer AG, Guerbet; A. Radbruch: personal fees for consulting and talks (within the last 3 years): Bayer, Guerbet, Novartis; financial study support: Bayer, Guerbet.
Figures


References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

