Macrophage-mediated anti-tumor immunity against high-risk neuroblastoma
- PMID: 35525858
- PMCID: PMC9232393
- DOI: 10.1038/s41435-022-00172-w
Macrophage-mediated anti-tumor immunity against high-risk neuroblastoma
Abstract
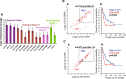
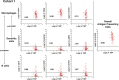
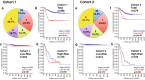
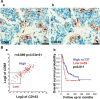
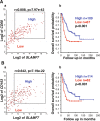
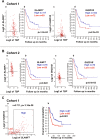
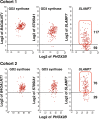
Neuroblastoma is the most common extracranial childhood solid tumor. The majority of high-risk neuroblastoma is resistant/refractory to the current high intensity therapy. Neuroblastoma lacks classical HLA Class I expression and exhibits low mutation burden, allowing neuroblastoma cells to evade CD8+ T cell-mediated immunity. Neuroblastoma cells do not express PD-L1, and tumor-associated macrophages are the predominant PD-L1+ cells in the tumor. In this study, we performed gene expression profiling and survival analyses on large neuroblastoma datasets to address the prognostic effect of PD-L1 gene expression and the possible involvement of the SLAMF7 pathway in the anti-neuroblastoma immunity. High-level expression of PD-L1 was found significantly associated with better outcome of high-risk neuroblastoma patients; two populations of PD-1+ PD-L1+ macrophages could be present in high-risk tumors with PD-1/PD-L1 ratios, ≈1 and >1. Patients with the PD-1/PD-L1 ratio >1 tumor showed inferior survival. High-level co-expression of SLAMF7 and SH2D1B was significantly associated with better survival of the high-risk neuroblastoma patients. Together, this study supports the hypothesis that macrophages are important effector cells in the anti-high-risk neuroblastoma immunity, that PD-1 blockade therapy can be beneficial to the high-risk neuroblastoma subset with the PD-1/PD-L1 expression ratio >1, and that SLAMF7 is a new therapeutic target of high-risk neuroblastoma.
© 2022. The Author(s).
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
PD-L1 Is a Therapeutic Target of the Bromodomain Inhibitor JQ1 and, Combined with HLA Class I, a Promising Prognostic Biomarker in Neuroblastoma.Clin Cancer Res. 2017 Aug 1;23(15):4462-4472. doi: 10.1158/1078-0432.CCR-16-2601. Epub 2017 Mar 7. Clin Cancer Res. 2017. PMID: 28270499
-
PD-L1-expressing natural killer cells predict favorable prognosis and response to PD-1/PD-L1 blockade in neuroblastoma.Oncoimmunology. 2023 Dec 1;13(1):2289738. doi: 10.1080/2162402X.2023.2289738. eCollection 2024. Oncoimmunology. 2023. PMID: 38125723 Free PMC article.
-
PD-L1 checkpoint inhibition and anti-CTLA-4 whole tumor cell vaccination counter adaptive immune resistance: A mouse neuroblastoma model that mimics human disease.PLoS Med. 2018 Jan 29;15(1):e1002497. doi: 10.1371/journal.pmed.1002497. eCollection 2018 Jan. PLoS Med. 2018. PMID: 29377881 Free PMC article.
-
PD-L1, inflammation, non-coding RNAs, and neuroblastoma: Immuno-oncology perspective.Semin Cancer Biol. 2018 Oct;52(Pt 2):53-65. doi: 10.1016/j.semcancer.2017.11.009. Epub 2017 Nov 28. Semin Cancer Biol. 2018. PMID: 29196189 Free PMC article. Review.
-
A Systematic Review of the Tumor-Infiltrating CD8+ T-Cells/PD-L1 Axis in High-Grade Glial Tumors: Toward Personalized Immuno-Oncology.Front Immunol. 2021 Sep 17;12:734956. doi: 10.3389/fimmu.2021.734956. eCollection 2021. Front Immunol. 2021. PMID: 34603316 Free PMC article.
Cited by
-
Identification and verification of international neuroblastoma staging system (INSS) stage-related genes as potential biomarkers for neuroblastoma prognostic models.Front Cell Dev Biol. 2025 Apr 15;13:1502380. doi: 10.3389/fcell.2025.1502380. eCollection 2025. Front Cell Dev Biol. 2025. PMID: 40302936 Free PMC article.
-
A Perspective on the CD47-SIRPA Axis in High-Risk Neuroblastoma.Curr Oncol. 2024 Jun 1;31(6):3212-3226. doi: 10.3390/curroncol31060243. Curr Oncol. 2024. PMID: 38920727 Free PMC article. Review.
-
Monocyte and Macrophage in Neuroblastoma: Blocking Their Pro-Tumoral Functions and Strengthening Their Crosstalk with Natural Killer Cells.Cells. 2023 Mar 13;12(6):885. doi: 10.3390/cells12060885. Cells. 2023. PMID: 36980226 Free PMC article. Review.
-
Dual roles of HK3 in regulating the network between tumor cells and tumor-associated macrophages in neuroblastoma.Cancer Immunol Immunother. 2024 May 7;73(7):122. doi: 10.1007/s00262-024-03702-9. Cancer Immunol Immunother. 2024. PMID: 38714539 Free PMC article.
-
Targeting the myeloid microenvironment in neuroblastoma.J Exp Clin Cancer Res. 2023 Dec 13;42(1):337. doi: 10.1186/s13046-023-02913-9. J Exp Clin Cancer Res. 2023. PMID: 38087370 Free PMC article. Review.
References
-
- Whelan JP, Chatten J, Lampson LA. HLA class I and b2-microglobulin expression in frozen and formaldehyde-fixed paraffin sections of neuroblastoma tumors. Cancer Res. 1985;45:5976–83. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

