Development and content validation of patient-reported outcomes tools for ulcerative colitis and Crohn's disease in adults with moderate-to-severe disease
- PMID: 35525935
- PMCID: PMC9078017
- DOI: 10.1186/s12955-022-01975-1
Development and content validation of patient-reported outcomes tools for ulcerative colitis and Crohn's disease in adults with moderate-to-severe disease
Abstract
Background: Ulcerative colitis (UC) and Crohn's disease (CD) are associated with a range of symptoms that adversely affect health-related quality of life. This research aimed to develop and validate two patient-reported outcome (PRO) tools to assess signs and symptoms in patients with moderate-to-severe UC or CD.
Methods: PRO-UC and PRO-CD Diaries were developed in accordance with US Food and Drug Administration (FDA) recommendations. Data were collected from concept elicitation interviews (in which patients described their symptoms and experience of the disease) and further refined through cognitive interviews (in which patients assessed the relevance and clarity of questions in the tools).
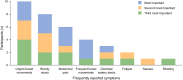
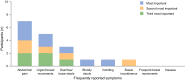
Results: Interviews were conducted with 12 patients for each indication. Five symptoms (urgent bowel movements, abdominal pain, frequent bowel movements, bloody stools, diarrhea/watery stools) were reported by 83-100% of participants with UC and were included in the final 6-item PRO-UC Diary: stool frequency, rectal bleeding (2 items), diarrhea, rectal urgency, and abdominal pain. For CD, seven symptoms (abdominal pain, diarrhea/loose stools, urgent bowel movements, fatigue/tiredness/weakness, frequent bowel movements, bloody stools, nausea) were reported by 50-100% of participants. These, together with vomiting and incontinence (reported by 42% and 33% of participants, respectively), were included in the final 10-item PRO-CD Diary, covering abdominal pain (2 items), stool frequency, liquid/very soft stool frequency, rectal bleeding, rectal urgency, nausea, vomiting, bowel incontinence, and general well-being. Symptoms were consistently cited across both indications to have an impact on quality of life, with frequent complaints being the need to always be near a toilet and inability to leave home, as well as general pain, discomfort, and nausea. For both tools, questions were accurately interpreted, with at least 67% of participants in both indications stating that items were easy to answer/relevant to their condition and symptoms were easy to recall over the last 24 h.
Conclusions: Both the PRO-UC and PRO-CD Diaries were developed and validated in accordance with FDA recommendations, providing two new tools for use in clinical trials to assess response to treatment in patients with UC or CD. Psychometric analyses are warranted to fully evaluate their properties and value for use in clinical trials.
Keywords: Crohn’s disease; Health-related quality of life; Inflammatory bowel disease; Patient-reported outcomes; Ulcerative colitis.
© 2022. The Author(s).
Conflict of interest statement
AN, currently employed at Sanofi, was an employee of Takeda Development Center Americas at the time of the study, and holds stock in Takeda. BR is a salaried employee of ICON, which received research funds from Shire, a Takeda company for conducting the study.
Figures
Similar articles
-
A patient-reported outcome measure comprising the stool frequency and abdominal pain items from the Crohn's Disease Activity Index: psychometric evaluation in adults with Crohn's disease.J Patient Rep Outcomes. 2025 Feb 17;9(1):19. doi: 10.1186/s41687-025-00851-y. J Patient Rep Outcomes. 2025. PMID: 39962027 Free PMC article. Clinical Trial.
-
Patient perception of bowel urgency and remission in moderately to severely active Crohn's disease or ulcerative colitis: a qualitative study.J Patient Rep Outcomes. 2024 Nov 12;8(1):130. doi: 10.1186/s41687-024-00800-1. J Patient Rep Outcomes. 2024. PMID: 39531103 Free PMC article.
-
Patient Perspectives of Bowel Urgency and Bowel Urgency-Related Accidents in Ulcerative Colitis and Crohn's Disease.Inflamm Bowel Dis. 2024 Oct 3;30(10):1831-1842. doi: 10.1093/ibd/izae044. Inflamm Bowel Dis. 2024. PMID: 38513272 Free PMC article.
-
Patient experiences in ulcerative colitis: conceptual model and review of patient-reported outcome measures.Qual Life Res. 2024 May;33(5):1373-1387. doi: 10.1007/s11136-024-03612-4. Epub 2024 Mar 5. Qual Life Res. 2024. PMID: 38438664 Free PMC article. Review.
-
A qualitative study to explore the symptoms and impacts of Crohn's disease and to develop the Crohn's Disease Diary.Qual Life Res. 2023 Jan;32(1):209-223. doi: 10.1007/s11136-022-03233-9. Epub 2022 Sep 2. Qual Life Res. 2023. PMID: 36056191 Free PMC article. Review.
Cited by
-
Disease Activity and Health-related Quality of Life Relationships with Work Productivity in Patients with Ulcerative Colitis in OCTAVE Induction 1 and 2 and OCTAVE Sustain.J Crohns Colitis. 2023 Apr 19;17(4):513-523. doi: 10.1093/ecco-jcc/jjac161. J Crohns Colitis. 2023. PMID: 36271912 Free PMC article. Clinical Trial.
-
A patient-reported outcome measure comprising the stool frequency and abdominal pain items from the Crohn's Disease Activity Index: psychometric evaluation in adults with Crohn's disease.J Patient Rep Outcomes. 2025 Feb 17;9(1):19. doi: 10.1186/s41687-025-00851-y. J Patient Rep Outcomes. 2025. PMID: 39962027 Free PMC article. Clinical Trial.
-
Bowel Urgency in Ulcerative Colitis: Current Perspectives and Future Directions.Am J Gastroenterol. 2023 Nov 1;118(11):1940-1953. doi: 10.14309/ajg.0000000000002404. Epub 2023 Jun 12. Am J Gastroenterol. 2023. PMID: 37436151 Free PMC article. Review.
-
Patient perception of bowel urgency and remission in moderately to severely active Crohn's disease or ulcerative colitis: a qualitative study.J Patient Rep Outcomes. 2024 Nov 12;8(1):130. doi: 10.1186/s41687-024-00800-1. J Patient Rep Outcomes. 2024. PMID: 39531103 Free PMC article.
-
Long-Term Improvement in the Patient-Reported Outcomes of Rectal Bleeding, Stool Frequency, and Health-Related Quality of Life with Tofacitinib in the Ulcerative Colitis OCTAVE Clinical Program.Patient. 2023 Mar;16(2):95-103. doi: 10.1007/s40271-022-00603-w. Epub 2022 Nov 7. Patient. 2023. PMID: 36336750 Free PMC article. Clinical Trial.
References
MeSH terms
LinkOut - more resources
Full Text Sources
Medical