Characterization of Inflammatory Factors and T Cell Subpopulations in a Murine Model of Kawasaki Disease Induced by Candida albicans Cell Wall Extracts (CAWS)
- PMID: 35526108
- PMCID: PMC9097462
- DOI: 10.12659/MSM.936355
Characterization of Inflammatory Factors and T Cell Subpopulations in a Murine Model of Kawasaki Disease Induced by Candida albicans Cell Wall Extracts (CAWS)
Abstract
Background: Kawasaki disease (KD) is an acute vasculitis in children, which ranks as the main cause of acquired heart disease in children in developed countries. The etiopathogenesis of KD remains to be clarified. Our study constructed a KD murine model and monitored the alterations of inflammatory factors and T cell subpopulations.
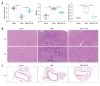
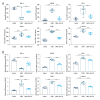
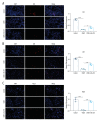
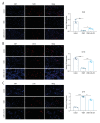
Material/Methods: Candida albicans cell wall extracts (CAWS) were utilized for inducing KD murine models. After 5 days, the mice were subcutaneously injected with rhG-CSF lasting 5 consecutive days. At 4 weeks, histopathology of hearts and arteries was observed via H&E staining. Inflammatory cytokine, chemokine, and adhesion molecule expression in serum specimens was detected via RT-qPCR and ELISA. T cell subpopulations in cardiac tissues were labeled through immunofluorescence and flow cytometry.
Results: CAWS induced cardiac dysfunction (reduced fraction shortening, increased left ventricle end-diastolic diameter and heart weight/body weight ratio) and cardiac and vascular inflammation, which were ameliorated by rhG-CSF treatment. CAWS-induced mice had increased levels of pro-inflammatory cytokines (IL-1β, IL-6, TNF-α), chemokine (RANTES), and adhesion molecules (MCP-1, VCAM-1, ICAM-1, E-selectin), as well as the decreased levels of anti-inflammatory cytokines (IL-10, G-CSF). Their levels were decreased following rhG-CSF administration. There were decreased CD3+, CD4+, CD8+, CD8+CD45+, Foxp3+, CD25+ and Foxp3+CD25+ T lymphocyte subpopulations and an increased CD45+ T lymphocyte subpopulation in cardiac tissues of CAWS-induced mice, which were ameliorated by rhG-CSF administration.
Conclusions: Our findings reveal the characteristics of inflammatory factors and T cell subpopulations in CAWS-induced KD. Moreover, rhG-CSF might be an effective therapeutic regimen against KD.
Conflict of interest statement
Figures



Similar articles
-
[Histopathological examination and analysis of mortality in DBA/2 mouse vasculitis induced with CAWS, a water-soluble extracellular polysaccharide fraction obtained from Candida albicans].Yakugaku Zasshi. 2006 Aug;126(8):643-50. doi: 10.1248/yakushi.126.643. Yakugaku Zasshi. 2006. PMID: 16880722 Japanese.
-
Murine model of Kawasaki disease induced by mannoprotein-beta-glucan complex, CAWS, obtained from Candida albicans.Jpn J Infect Dis. 2004 Oct;57(5):S9-10. Jpn J Infect Dis. 2004. PMID: 15507772
-
Candesartan Attenuates Vasculitis in a Mouse Model of Kawasaki Disease Induced by Candida albicans Water-Soluble Fraction.J Nippon Med Sch. 2024;91(3):285-295. doi: 10.1272/jnms.JNMS.2024_91-307. J Nippon Med Sch. 2024. PMID: 38972741
-
[Coronary arteritis induced by CAWS (Candida albicans water-soluble fraction) in various strains of mice].Nihon Ishinkin Gakkai Zasshi. 2008;49(4):287-92. doi: 10.3314/jjmm.49.287. Nihon Ishinkin Gakkai Zasshi. 2008. PMID: 19001755 Review. Japanese.
-
Molecular organization of the cell wall of Candida albicans.Med Mycol. 2001;39 Suppl 1:1-8. Med Mycol. 2001. PMID: 11800263 Review.
Cited by
-
Impact of COVID-19 infection on Kawasaki disease and immune status in children.Sci Rep. 2025 Feb 21;15(1):6417. doi: 10.1038/s41598-025-91042-8. Sci Rep. 2025. PMID: 39984588 Free PMC article.
References
-
- Koné-Paut I, Tellier S, Belot A, et al. Phase II open label study of anakinra in intravenous immunoglobulin-resistant Kawasaki disease. Arthritis Rheumatol. 2021;73:151–61. - PubMed
-
- Liu E, Gonzalez J, Siu A. Use of premedication with intravenous immune globulin in Kawasaki disease: A retrospective review. Pediatr Allergy Immunol. 2021;32:750–55. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

