Altered Vagal Signaling and Its Pathophysiological Roles in Functional Dyspepsia
- PMID: 35527812
- PMCID: PMC9072791
- DOI: 10.3389/fnins.2022.858612
Altered Vagal Signaling and Its Pathophysiological Roles in Functional Dyspepsia
Abstract
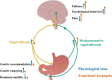
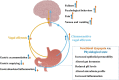
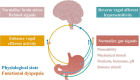
The vagus nerve is crucial in the bidirectional communication between the gut and the brain. It is involved in the modulation of a variety of gut and brain functions. Human studies indicate that the descending vagal signaling from the brain is impaired in functional dyspepsia. Growing evidence indicate that the vagal signaling from gut to brain may also be altered, due to the alteration of a variety of gut signals identified in this disorder. The pathophysiological roles of vagal signaling in functional dyspepsia is still largely unknown, although some studies suggested it may contribute to reduced food intake and gastric motility, increased psychological disorders and pain sensation, nausea and vomiting. Understanding the alteration in vagal signaling and its pathophysiological roles in functional dyspepsia may provide information for new potential therapeutic treatments of this disorder. In this review, we summarize and speculate possible alterations in vagal gut-to-brain and brain-to-gut signaling and the potential pathophysiological roles in functional dyspepsia.
Keywords: functional dyspepsia; gut–brain axis; vagal afferent; vagal efferent; vagus nerve.
Copyright © 2022 Li and Page.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Transcutaneous auricular vagal nerve stimulation improves functional dyspepsia by enhancing vagal efferent activity.Am J Physiol Gastrointest Liver Physiol. 2021 May 1;320(5):G700-G711. doi: 10.1152/ajpgi.00426.2020. Epub 2021 Feb 24. Am J Physiol Gastrointest Liver Physiol. 2021. PMID: 33624527 Free PMC article.
-
Vagal gut-brain signaling mediates amygdaloid plasticity, affect, and pain in a functional dyspepsia model.JCI Insight. 2021 Mar 22;6(6):e144046. doi: 10.1172/jci.insight.144046. JCI Insight. 2021. PMID: 33591956 Free PMC article.
-
Meal-Sensing Signaling Pathways in Functional Dyspepsia.Front Syst Neurosci. 2018 Apr 5;12:10. doi: 10.3389/fnsys.2018.00010. eCollection 2018. Front Syst Neurosci. 2018. PMID: 29674959 Free PMC article. Review.
-
Recognizing the role of the vagus nerve in depression from microbiota-gut brain axis.Front Neurol. 2022 Nov 10;13:1015175. doi: 10.3389/fneur.2022.1015175. eCollection 2022. Front Neurol. 2022. PMID: 36438957 Free PMC article. Review.
-
Cine gastric MRI reveals altered Gut-Brain Axis in Functional Dyspepsia: gastric motility is linked with brainstem-cortical fMRI connectivity.Neurogastroenterol Motil. 2022 Oct;34(10):e14396. doi: 10.1111/nmo.14396. Epub 2022 May 13. Neurogastroenterol Motil. 2022. PMID: 35560690 Free PMC article.
Cited by
-
Acupuncture improves the symptoms, serum ghrelin, and autonomic nervous system of patients with postprandial distress syndrome: a randomized controlled trial.Chin Med. 2024 Nov 20;19(1):162. doi: 10.1186/s13020-024-01028-3. Chin Med. 2024. PMID: 39568071 Free PMC article.
-
Perianaesthetic challenges in patients undergoing vagus nerve stimulation (VNS) placement.BMJ Case Rep. 2023 Feb 7;16(2):e252692. doi: 10.1136/bcr-2022-252692. BMJ Case Rep. 2023. PMID: 36750294 Free PMC article.
-
Pregnancy and a high-fat, high-sugar diet each attenuate mechanosensitivity of murine gastric vagal afferents, with no additive effects.J Physiol. 2025 Mar;603(6):1461-1481. doi: 10.1113/JP286115. Epub 2025 Mar 2. J Physiol. 2025. PMID: 40023799 Free PMC article.
-
Exploring the Connection between Sleep Disorders, Emotional Distress, and Quality of Life in Functional Dyspepsia.Actas Esp Psiquiatr. 2025 Mar;53(2):222-229. doi: 10.62641/aep.v53i2.1947. Actas Esp Psiquiatr. 2025. PMID: 40071357 Free PMC article.
-
Investigating the modulation of gastric sensations and disposition toward food with taVNS.Psychophysiology. 2025 Feb;62(2):e14735. doi: 10.1111/psyp.14735. Epub 2024 Nov 30. Psychophysiology. 2025. PMID: 39614658 Free PMC article.
References
-
- Abbott C. R., Monteiro M., Small C. J., Sajedi A., Smith K. L., Parkinson J. R., et al. (2005). The inhibitory effects of peripheral administration of peptide YY(3-36) and glucagon-like peptide-1 on food intake are attenuated by ablation of the vagal-brainstem-hypothalamic pathway. Brain Res. 1044 127–131. 10.1016/j.brainres.2005.03.011 - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources

