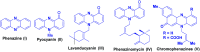
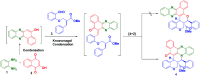
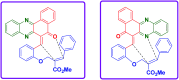
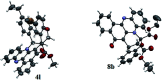
Two step, one-pot sequential synthesis of functionalized hybrid polyheterocyclic scaffolds via a solid state melt reaction (SSMR)
- PMID: 35527912
- PMCID: PMC9069754
- DOI: 10.1039/c9ra02590a
Two step, one-pot sequential synthesis of functionalized hybrid polyheterocyclic scaffolds via a solid state melt reaction (SSMR)
Abstract
A new one pot assembly of highly functionalized benzo[a]phenazinone fused chromene/bicyclic scaffolds via a domino Knoevenagel intramolecular hetero-Diels-Alder (IMHDA) strategy using a solid state melt reaction (SSMR) of 2-hydroxynaphthalene 1,4-dione, o-phenylenediamine, O-allyl salicylaldehyde/O-vinyl salicylaldehyde derivatives is reported. The formation of five new bonds (two C-C bonds and three C-O bonds), three six-membered rings, and three stereogenic centers in a one-pot manner is very attractive. Ease of reaction with short time, good yields with water as the only byproduct and work up free procedure are some of the excellent features of the present protocol.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures
Similar articles
-
Construction of hybrid polycyclic quinolinobenzo[a]phenazinone architectures using solid-state melt reaction (SSMR).Mol Divers. 2021 Nov;25(4):2447-2458. doi: 10.1007/s11030-020-10090-6. Epub 2020 May 4. Mol Divers. 2021. PMID: 32367313
-
Synthesis of fused uracils: pyrano[2,3-d]pyrimidines and 1,4-bis(pyrano[2,3-d]pyrimidinyl)benzenes by domino Knoevenagel/Diels-Alder reactions.Monatsh Chem. 2012;143(8):1175-1185. doi: 10.1007/s00706-012-0781-x. Epub 2012 May 24. Monatsh Chem. 2012. PMID: 26166870 Free PMC article.
-
One-Pot Double-Annulation Strategy for the Synthesis of Unusual Fused Bis-Heterocycles.Org Lett. 2020 Jun 5;22(11):4350-4354. doi: 10.1021/acs.orglett.0c01351. Epub 2020 May 15. Org Lett. 2020. PMID: 32412769
-
BF₃·OEt₂ mediated metal-free one-pot sequential multiple annulation cascade (SMAC) synthesis of complex and diverse tetrahydroisoquinoline fused hybrid molecules.Org Biomol Chem. 2016 Mar 28;14(12):3207-11. doi: 10.1039/c6ob00253f. Org Biomol Chem. 2016. PMID: 26935814
-
A domino Knoevenagel hetero-Diels-Alder reaction for the synthesis of polycyclic chromene derivatives and evaluation of their cytotoxicity.Bioorg Med Chem Lett. 2012 Mar 1;22(5):1995-9. doi: 10.1016/j.bmcl.2012.01.033. Epub 2012 Jan 21. Bioorg Med Chem Lett. 2012. PMID: 22330634
References
-
-
For some reviews on MC reactions, see:
- Armstrong R. M. Combs A. P. Brown P. A. Keating T. A. Acc. Chem. Res. 1996;29:123–131. doi: 10.1021/ar9502083. - DOI
- Dömling A. Ugi I. Angew. Chem., Int. Ed. 2000;39:3169–3210. doi: 10.1002/1521-3773(20000915)39:18<3168::AID-ANIE3168>3.0.CO;2-U. - DOI - PubMed
- Bienaymé H. Hulme C. Oddon G. Schmitt P. Chem. –Eur. J. 2000;6:3321–3329. doi: 10.1002/1521-3765(20000915)6:18<3321::AID-CHEM3321>3.0.CO;2-A. - DOI - PubMed
- Dömling A. Curr. Opin. Chem. Biol. 2000;4:318–323. doi: 10.1016/S1367-5931(00)00095-8. - DOI - PubMed
- Dömling A. Chem. Rev. 2006;106:17–89. doi: 10.1021/cr0505728. - DOI - PubMed
- Isambert N. Lavilla R. Chem. –Eur. J. 2008;14:8444–8454. doi: 10.1002/chem.200800473. - DOI - PubMed
- Ganem B. Acc. Chem. Res. 2009;42:463–472. doi: 10.1021/ar800214s. - DOI - PMC - PubMed
- Ruijter E. Scheffelaar R. Orru R. V. Angew. Chem., Int. Ed. 2011;50:6234–6246. doi: 10.1002/anie.201006515. - DOI - PubMed
-
-
- Andraos J. Org. Process Res. Dev. 2005;9:149–163. doi: 10.1021/op049803n. - DOI
-
- Dictionary of Alkaloids, ed., I. W.Southon and J.Buckingham, Chapman & Hall, New York, 1989
-
- Laursen J. B. Nielsen J. Chem. Rev. 2004;104:1663–1686. doi: 10.1021/cr020473j. - DOI - PubMed
- Makgatho M. Anderson R. O'Sullivan J. Egan T. Freese J. Cornelius N. Van Rensburg C. Drug Dev. Res. 2000;50:195–202. doi: 10.1002/1098-2299(200006)50:2<195::AID-DDR10>3.0.CO;2-T. - DOI
- Andrade-Nieto V. Goulart M. da Silva J. F. da Silva M. J. Pinto M. Pinto A. Zalis M. Carvalho L. Krettli A. Bioorg. Med. Chem. Lett. 2004;14:1145–1149. doi: 10.1016/j.bmcl.2003.12.069. - DOI - PubMed
- Neves-Pinto C. Malta V. Pinto M. Santos R. Castro S. Pinto A. J. Med. Chem. 2002;45:740–743. doi: 10.1021/jm010377v. - DOI - PubMed
- Cartwright D. Chilton W. Benson D. Appl. Microbiol. Biotechnol. 1995;43:211–216. doi: 10.1007/BF00172814. - DOI
- Ligon J. Dwight S. Hammer P. Torkewitz N. Hofmann D. Kempf H. Pee K. Pest Manage. Sci. 2000;56:688–695. doi: 10.1002/1526-4998(200008)56:8<688::AID-PS186>3.0.CO;2-V. - DOI
- Muller M. Sorrell T. Prostaglandins. 1995;50:301–311. doi: 10.1016/0090-6980(95)00133-6. - DOI - PubMed
LinkOut - more resources
Full Text Sources