Use of Continuous Glucose Monitoring in the Assessment and Management of Patients With Diabetes and Chronic Kidney Disease
- PMID: 35528010
- PMCID: PMC9074296
- DOI: 10.3389/fendo.2022.869899
Use of Continuous Glucose Monitoring in the Assessment and Management of Patients With Diabetes and Chronic Kidney Disease
Abstract
In developed countries, diabetes is the leading cause of chronic kidney disease (CKD) and accounts for 50% of incidence of end stage kidney disease. Despite declining prevalence of micro- and macrovascular complications, there are rising trends in renal replacement therapy in diabetes. Optimal glycemic control may reduce risk of progression of CKD and related death. However, assessing glycemic control in patients with advanced CKD and on dialysis (G4-5) can be challenging. Laboratory biomarkers, such as glycated haemoglobin (HbA1c), may be biased by abnormalities in blood haemoglobin, use of iron therapy and erythropoiesis-stimulating agents and chronic inflammation due to uraemia. Similarly, glycated albumin and fructosamine may be biased by abnormal protein turnover. Patients with advanced CKD exhibited heterogeneity in glycemic control ranging from severe insulin resistance to 'burnt-out' beta-cell function. They also had high risk of hypoglycaemia due to reduced renal gluconeogenesis, frequent use of insulin and dysregulation of counterregulatory hormones. Continuous glucose monitoring (CGM) systems measure glucose in interstitial fluid every few minutes and provide an alternative and more reliable method of glycemic assessment, including asymptomatic hypoglycaemia and hyperglycaemic excursions. Recent international guidelines recommended use of CGM-derived Glucose Management Index (GMI) in patients with advanced CKD although data are scarce in this population. Using CGM, patients with CKD were found to experience marked glycemic fluctuations with hypoglycemia due to loss of glucose and insulin during haemodialysis (HD) followed by hyperglycemia in the post-HD period. On the other hand, during peritoneal dialysis, patients may experience glycemic excursions with influx of glucose from dialysate solutions. These undesirable glucose exposure and variability may accelerate decline of residual renal function. Although CGM may improve the quality of glycemic monitoring and control in populations with CKD, further studies are needed to confirm the accuracy, optimal mode and frequency of CGM as well as their cost-effectiveness and user-acceptability in patients with advanced CKD and dialysis.
Keywords: continuous glucose monitoring; diabetes; diabetic kidney disease; diabetic nephropathy; dialysis; end stage kidney disease (ESKD); type 2 (non-insulin-dependent) diabetes mellitus.
Copyright © 2022 Ling, Ng, Chan and Chow.
Conflict of interest statement
EC has received institutional research support form Medtronic Diabetes and Powder Pharmaceuticals Inc. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
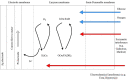
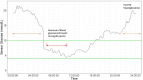
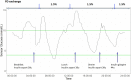
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

