Graphene oxide-based electrochemical activation of ethionamide towards enhanced biological activity
- PMID: 35528088
- PMCID: PMC9074427
- DOI: 10.1039/c9ra06681k
Graphene oxide-based electrochemical activation of ethionamide towards enhanced biological activity
Abstract
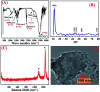
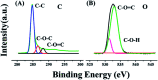
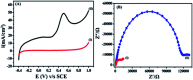
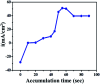
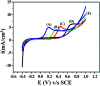
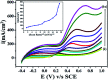
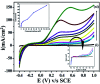
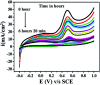
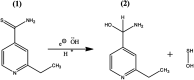
The electrochemical behavior of ethionamide (ETO) was investigated on GO (∼500 nm) using the linear sweep voltammetric (LSV) technique at the sweep rate of 10 mV s-1 in 1 M PBS buffer solution, and the characteristic anodic signal was examined at 0.240 V over the potential range of -0.4 to 1 V vs. SCE. However, linearity was observed with the increase in scan rate (2-300 mV s-1) and concentration of ETO (1 μM to 100 mM), suggesting that the process involved diffusion-controlled electron transfer. The results also exhibited excellent current and potential stability, limit of detection (LOD 1.33) and limit of quantification (LOQ 4.4) at optimized experimental conditions. This electrochemical oxidation method was successfully applied in the complete oxidation of ETO to its oxidized form, which was further confirmed by high resolution mass spectroscopy (HRMS) and Fourier transform infrared (FTIR) spectroscopic measurements. Interestingly, the comparative biological evaluation of ETO and ETO-O (oxidised form) showed good enhancement in the activity of oxidised ETO against some Gram-negative pathogens, such as E. aerogenes, S. abony, S. boydii, and E. coli.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures
Similar articles
-
Green synthesis of silver nanoparticles-graphene oxide nanocomposite and its application in electrochemical sensing of tryptophan.Biosens Bioelectron. 2013 Apr 15;42:198-206. doi: 10.1016/j.bios.2012.10.029. Epub 2012 Oct 26. Biosens Bioelectron. 2013. PMID: 23202352
-
High-sensitivity paracetamol sensor based on Pd/graphene oxide nanocomposite as an enhanced electrochemical sensing platform.Biosens Bioelectron. 2014 Apr 15;54:468-75. doi: 10.1016/j.bios.2013.11.001. Epub 2013 Nov 8. Biosens Bioelectron. 2014. PMID: 24315879
-
Synthesis and characterization of the thermally reduced graphene oxide in argon atmosphere, and its application to construct graphene paste electrode as a naptalam electrochemical sensor.Anal Chim Acta. 2018 Dec 4;1035:22-31. doi: 10.1016/j.aca.2018.06.057. Epub 2018 Jun 21. Anal Chim Acta. 2018. PMID: 30224141
-
Enhancement effect of reduced graphene oxide and silver nanocomposite supported on poly brilliant blue platform for ultra-trace voltammetric analysis of rosuvastatin in tablets and human plasma.RSC Adv. 2019 Mar 1;9(13):7136-7146. doi: 10.1039/c8ra10415h. eCollection 2019 Mar 1. RSC Adv. 2019. PMID: 35519976 Free PMC article.
-
A Laser Reduced Graphene Oxide Grid Electrode for the Voltammetric Determination of Carbaryl.Molecules. 2021 Aug 20;26(16):5050. doi: 10.3390/molecules26165050. Molecules. 2021. PMID: 34443639 Free PMC article.
Cited by
-
Combination of CNTs with Classical Drugs for Treatment in Human Colorectal Adenocarcinoma (HT-29) Cell Line.Nanomaterials (Basel). 2023 Jun 25;13(13):1933. doi: 10.3390/nano13131933. Nanomaterials (Basel). 2023. PMID: 37446448 Free PMC article.
References
-
- WHO Report 2012, World Health Organization, Geneva, Switzerland, 2012, Global TB report, 2016
-
- Broaddus V. C., Mason R. J., Ernst J. D., King T. E., Lazarus S. C., Murray J. F., Nadel J. A., Slutsky A. S. and Gotway M. B., Tuberculosis, Murray and Nadel's Textbook of Respiratory Medicine, 6th edn, 2016, p. 628
-
- Global tuberculosis report 2018, Archived from the original on Sept. 2018
-
- Sarkar S. Ganguly A. Sunwoo H. H. Mol. Microbiol. 2016;6:2–8.
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

