Rotary manifold for automating a paper-based Salmonella immunoassay
- PMID: 35528425
- PMCID: PMC9071810
- DOI: 10.1039/c9ra07106g
Rotary manifold for automating a paper-based Salmonella immunoassay
Abstract
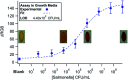
Foodborne pathogens are responsible for hundreds of thousands of deaths around the world each year. Rapid screening of agricultural products for these pathogens is essential to reduce and/or prevent outbreaks and pinpoint contamination sources. Unfortunately, current detection methods are laborious, expensive, time-consuming and require a central laboratory. Therefore, a rapid, sensitive, and field-deployable pathogen-detection assay is needed. We previously developed a colorimetric sandwich immunoassay utilizing immuno-magnetic separation (IMS) and chlorophenol red-β-d-galactopyranoside for Salmonella detection on a paper-based analytical device (μPAD); however, the assay required many sample preparation steps prior to the μPAD as well as laboratory equipment, which decreased user-friendliness for future end-users. As a step towards overcoming these limitations in resource-limited settings, we demonstrate a reusable 3D-printed rotational manifold that couples with disposable μPAD layers for semi-automated reagent delivery, washing, and detection in 65 minutes. After IMS to clean the sample, the manifold performs pipette-free reagent delivery and washing steps in a sequential order with controlled volumes, followed by enzymatic amplification and colorimetric detection using automated image processing to quantify color change. Salmonella was used as the target pathogen in this project and was detected with the manifold in growth media and milk with detection limits of 4.4 × 102 and 6.4 × 102 CFU mL-1 respectively. The manifold increases user friendliness and simplifies immunoassays resulting in a practical product for in-field use and commercialization.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures





Similar articles
-
Highly Sensitive Detection of Salmonella typhimurium Using a Colorimetric Paper-Based Analytical Device Coupled with Immunomagnetic Separation.Anal Chem. 2018 Jan 2;90(1):1035-1043. doi: 10.1021/acs.analchem.7b04628. Epub 2017 Dec 20. Anal Chem. 2018. PMID: 29211962
-
Magnetic Bead Handling Using a Paper-Based Device for Quantitative Point-of-Care Testing.Biosensors (Basel). 2022 Aug 25;12(9):680. doi: 10.3390/bios12090680. Biosensors (Basel). 2022. PMID: 36140066 Free PMC article.
-
Rapid Bacteria Detection at Low Concentrations Using Sequential Immunomagnetic Separation and Paper-Based Isotachophoresis.Anal Chem. 2019 Aug 6;91(15):9623-9630. doi: 10.1021/acs.analchem.9b01002. Epub 2019 Jul 18. Anal Chem. 2019. PMID: 31282665
-
A rapid colorimetric immunoassay for the detection of pathogenic bacteria on poultry processing plants using cotton swabs and nanobeads.Mikrochim Acta. 2018 Feb 10;185(3):164. doi: 10.1007/s00604-018-2696-7. Mikrochim Acta. 2018. PMID: 29594804
-
Microfluidic devices for sample preparation and rapid detection of foodborne pathogens.Biotechnol Adv. 2018 Jul-Aug;36(4):1003-1024. doi: 10.1016/j.biotechadv.2018.03.002. Epub 2018 Mar 10. Biotechnol Adv. 2018. PMID: 29534915 Review.
Cited by
-
Capillary flow-driven immunoassay platform for COVID-19 antigen diagnostics.Anal Chim Acta. 2023 Oct 9;1277:341634. doi: 10.1016/j.aca.2023.341634. Epub 2023 Jul 27. Anal Chim Acta. 2023. PMID: 37604607 Free PMC article.
-
Paper-based sensors for bacteria detection.Nat Rev Bioeng. 2023;1(3):180-192. doi: 10.1038/s44222-023-00024-w. Epub 2023 Feb 14. Nat Rev Bioeng. 2023. PMID: 36937095 Free PMC article. Review.
-
Capillary driven microfluidic sequential flow device for point-of-need ELISA: COVID-19 serology testing.Anal Methods. 2023 Jun 8;15(22):2721-2728. doi: 10.1039/d3ay00225j. Anal Methods. 2023. PMID: 37099406 Free PMC article.
-
Recent Developments in Microfluidic Paper-based Analytical Devices for Pharmaceutical Analysis.Curr Top Med Chem. 2022;22(27):2241-2260. doi: 10.2174/1568026623666221027144310. Curr Top Med Chem. 2022. PMID: 36305123
-
Recent Advances in Microfluidic Devices for Contamination Detection and Quality Inspection of Milk.Micromachines (Basel). 2021 May 14;12(5):558. doi: 10.3390/mi12050558. Micromachines (Basel). 2021. PMID: 34068982 Free PMC article. Review.
References
-
- Group, F. D. B. E. R., WHO Estimates of the Global Burden of Foodborne Diseases, Organization, W. H., 2015
LinkOut - more resources
Full Text Sources
Research Materials

