Recent progress of solution-processed Cu nanowires transparent electrodes and their applications
- PMID: 35528598
- PMCID: PMC9070619
- DOI: 10.1039/c9ra04404c
Recent progress of solution-processed Cu nanowires transparent electrodes and their applications
Abstract
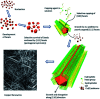
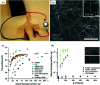
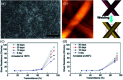
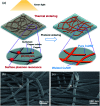
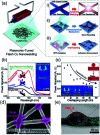
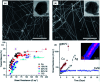
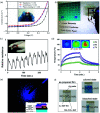
Research on next-generation transparent electrode (TE) materials to replace expensive and fragile indium tin oxide (ITO) is crucial for future electronics. Copper nanowires (Cu NWs) are considered as one of the most promising alternatives due to their excellent electrical properties and low-cost processing. This review summarizes the recent progress on the synthesis methods of long Cu NWs, and the fabrication techniques and protection measures for Cu NW TEs. Applications of Cu NW TEs in electronics, such as solar cells, touch screens, and light emitting diodes (LEDs), are discussed.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures






Similar articles
-
Nanowire-based transparent conductors for flexible electronics and optoelectronics.Sci Bull (Beijing). 2017 Jan 30;62(2):143-156. doi: 10.1016/j.scib.2016.11.009. Epub 2016 Dec 7. Sci Bull (Beijing). 2017. PMID: 36659486 Review.
-
Copper Nanowires and Their Applications for Flexible, Transparent Conducting Films: A Review.Nanomaterials (Basel). 2016 Mar 9;6(3):47. doi: 10.3390/nano6030047. Nanomaterials (Basel). 2016. PMID: 28344304 Free PMC article. Review.
-
Fast fabrication of copper nanowire transparent electrodes by a high intensity pulsed light sintering technique in air.Phys Chem Chem Phys. 2015 Dec 14;17(46):31110-6. doi: 10.1039/c5cp04582g. Phys Chem Chem Phys. 2015. PMID: 26536570
-
Low-temperature synthesis of indium tin oxide nanowires as the transparent electrodes for organic light emitting devices.Nanotechnology. 2012 Jan 20;23(2):025706. doi: 10.1088/0957-4484/23/2/025706. Nanotechnology. 2012. PMID: 22166812
-
Cost-Effective Fabrication of Uniformly Aligned Silver Nanowire Microgrid-Based Transparent Electrodes with Higher than 99% Transmittance.ACS Appl Mater Interfaces. 2022 Aug 31;14(34):39199-39210. doi: 10.1021/acsami.2c09672. Epub 2022 Aug 17. ACS Appl Mater Interfaces. 2022. PMID: 35976981
Cited by
-
Highly stretchable electroluminescent device based on copper nanowires electrode.Sci Rep. 2022 May 27;12(1):8967. doi: 10.1038/s41598-022-13167-4. Sci Rep. 2022. PMID: 35624312 Free PMC article.
-
Surface and Interface Designs in Copper-Based Conductive Inks for Printed/Flexible Electronics.Nanomaterials (Basel). 2020 Aug 27;10(9):1689. doi: 10.3390/nano10091689. Nanomaterials (Basel). 2020. PMID: 32867267 Free PMC article. Review.
-
Directly Printed Embedded Metal Mesh for Flexible Transparent Electrode via Liquid Substrate Electric-Field-Driven Jet.Adv Sci (Weinh). 2022 May;9(14):e2105331. doi: 10.1002/advs.202105331. Epub 2022 Mar 1. Adv Sci (Weinh). 2022. PMID: 35233960 Free PMC article.
References
Publication types
LinkOut - more resources
Full Text Sources

