Biglycan as a potential regulator of tumorgenicity and immunogenicity in K-RAS-transformed cells
- PMID: 35529675
- PMCID: PMC9067524
- DOI: 10.1080/2162402X.2022.2069214
Biglycan as a potential regulator of tumorgenicity and immunogenicity in K-RAS-transformed cells
Abstract
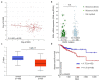
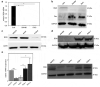
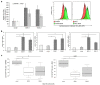
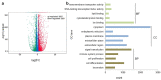
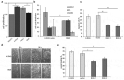
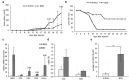
The extracellular matrix component biglycan (BGN) plays an essential role in various physiological and pathophysiological processes. A deficient BGN expression associated with reduced immunogenicity was found in HER-2/neu-overexpressing cells. To determine whether BGN is suppressed by oncogene-driven regulatory networks, the expression and function of BGN was analyzed in murine and human BGNlow/BGNhigh K-RASG12V-transformed model systems as well as in different patients' datasets of colorectal carcinoma (CRC) lesions. K-RAS-mutated CRC tissues expressed low BGN mRNA and protein levels when compared to normal colon epithelial cells, which was associated with a reduced patients' survival. Transfection of BGN in murine and human BGNlow K-RAS-expressing cells resulted in a reduced growth and migration of BGNhigh vs BGNlow K-RAS cells. In addition, increased MHC class I surface antigens as a consequence of an enhanced antigen processing machinery component expression was found upon restoration of BGN, which was confirmed by RNA-sequencing of BGNlow vs. BGNhigh K-RAS models. Furthermore, a reduced tumor formation of BGNhigh versus BGNlow K-RAS-transformed fibroblasts associated with an enhanced MHC class I expression and an increased frequency of tumor-infiltrating lymphocytes in tumor lesions was found. Our data provide for the first time an inverse link between BGN and K-RAS expression in murine and human K-RAS-overexpressing models and CRC lesions associated with altered growth properties, reduced immunogenicity and worse patients' outcome. Therefore, reversion of BGN might be a novel therapeutic option for K-RAS-associated malignancies.
Keywords: Immune escape; K-RAS; MHC class I; colorectal carcinoma; extracellular matrix protein; oncogene; proteoglycan.
© 2022 The Author(s). Published with license by Taylor & Francis Group, LLC.
Conflict of interest statement
No potential conflict of interest was reported by the author(s)
Figures

References
-
- Manou D, Caon I, Bouris P, Triantaphyllidou IE, Giaroni C, Passi A, Karamanos NK, Vigetti D, Theocharis AD. The complex interplay between extracellular matrix and cells in tissues. Methods Mol Biol. 2019;1952:1–20. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
