Development and Validation of a Prognostic Model to Predict Recurrence-Free Survival After Curative Resection for Perihilar Cholangiocarcinoma: A Multicenter Study
- PMID: 35530316
- PMCID: PMC9071302
- DOI: 10.3389/fonc.2022.849053
Development and Validation of a Prognostic Model to Predict Recurrence-Free Survival After Curative Resection for Perihilar Cholangiocarcinoma: A Multicenter Study
Abstract
Background: Recurrence is the main cause of death in perihilar cholangiocarcinoma (pCCA) patients after surgery. Identifying patients with a high risk of recurrence is important for decision-making regarding neoadjuvant therapy to improve long-term outcomes.
Aim: The objective of this study was to develop and validate a prognostic model to predict recurrence-free survival (RFS) after curative resection of pCCA.
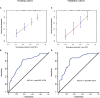
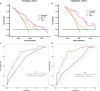
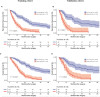
Methods: Patients following curative resection for pCCA from January 2008 to January 2016 were identified from a multicenter database. Using random assignment, 70% of patients were assigned to the training cohort, and the remaining 30% were assigned to the validation cohort. Independent predictors of RFS after curative resection for pCCA were identified and used to construct a prognostic model. The predictive performance of the model was assessed using calibration curves and the C-index.
Results: A total of 341 patients were included. The median overall survival (OS) was 22 months, and the median RFS was 14 months. Independent predictors associated with RFS included lymph node involvement, macrovascular invasion, microvascular invasion, maximum tumor size, tumor differentiation, and carbohydrate antigen 19-9. The model incorporating these factors to predict 1-year RFS demonstrated better calibration and better performance than the 8th American Joint Committee on Cancer (AJCC) staging system in both the training and validation cohorts (C-indexes: 0.723 vs. 0.641; 0.743 vs. 0.607).
Conclusions: The prognostic model could identify patients at high risk of recurrence for pCCA to inform patients and surgeons, help guide decision-making for postoperative adjuvant therapy, and improve survival.
Keywords: oncology; perihilar cholangiocarcinoma; prognostic model; recurrence; resection.
Copyright © 2022 Liu, Chen, Wang, Liu, Fan, Xu, Pan, Zhong, Xie, Bai, Jiang, Zhang, Dai and Chen.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Predictive model for very early recurrence of patients with perihilar cholangiocarcinoma: a machine learning approach.Hepatobiliary Surg Nutr. 2025 Feb 1;14(1):3-15. doi: 10.21037/hbsn-24-385. Epub 2025 Jan 16. Hepatobiliary Surg Nutr. 2025. PMID: 39925922 Free PMC article.
-
Development of a model based on the age-adjusted Charlson comorbidity index to predict survival for resected perihilar cholangiocarcinoma.World J Gastrointest Oncol. 2023 Jun 15;15(6):1036-1050. doi: 10.4251/wjgo.v15.i6.1036. World J Gastrointest Oncol. 2023. PMID: 37389112 Free PMC article.
-
Development and Assessment of a Clinical Calculator for Estimating the Likelihood of Recurrence and Survival Among Patients With Locally Advanced Rectal Cancer Treated With Chemotherapy, Radiotherapy, and Surgery.JAMA Netw Open. 2021 Nov 1;4(11):e2133457. doi: 10.1001/jamanetworkopen.2021.33457. JAMA Netw Open. 2021. PMID: 34748003 Free PMC article.
-
Systematic Review of Preoperative Prognostic Biomarkers in Perihilar Cholangiocarcinoma.Cancers (Basel). 2024 Feb 7;16(4):698. doi: 10.3390/cancers16040698. Cancers (Basel). 2024. PMID: 38398089 Free PMC article. Review.
-
Outcomes in biliary malignancy.J Surg Oncol. 2014 Oct;110(5):585-91. doi: 10.1002/jso.23762. J Surg Oncol. 2014. PMID: 25250887 Review.
Cited by
-
AI-based digital histopathology for perihilar cholangiocarcinoma: A step, not a jump.J Pathol Inform. 2023 Nov 5;15:100345. doi: 10.1016/j.jpi.2023.100345. eCollection 2024 Dec. J Pathol Inform. 2023. PMID: 38075015 Free PMC article.
-
Short- and long-term outcomes of laparoscopic versus open resection of perihilar cholangiocarcinoma: a propensity score-based analysis.Hepatobiliary Surg Nutr. 2025 Apr 1;14(2):207-221. doi: 10.21037/hbsn-23-680. Epub 2024 Aug 12. Hepatobiliary Surg Nutr. 2025. PMID: 40342757 Free PMC article.
-
Clinical features and prediction of long-term survival after surgery for perihilar cholangiocarcinoma.PLoS One. 2024 Jul 1;19(7):e0304838. doi: 10.1371/journal.pone.0304838. eCollection 2024. PLoS One. 2024. PMID: 38950006 Free PMC article.
-
Residual biliary intraepithelial neoplasia without malignant transformation at resection margin for perihilar cholangiocarcinoma does not require expanded resection: a dual center retrospective study.World J Surg Oncol. 2024 Jun 21;22(1):161. doi: 10.1186/s12957-024-03395-5. World J Surg Oncol. 2024. PMID: 38907218 Free PMC article.
-
Impact of perioperative blood transfusion on long-term survival in patients with different stages of perihilar cholangiocarcinoma treated with curative resection: A multicentre propensity score matching study.Front Oncol. 2022 Oct 31;12:1059581. doi: 10.3389/fonc.2022.1059581. eCollection 2022. Front Oncol. 2022. PMID: 36387093 Free PMC article.
References
LinkOut - more resources
Full Text Sources

